Document Type : Review Article
Authors
- Rupali C Shete 1
- Pearl Rhea Fernandes 2
- Bravish Raju Borhade 3
- Amit Anil Pawar 3
- Maryappa Chudappa Sonawane 4
- Nikhil sanjay warude 3
1 Department of Chemistry, CKT College Panvel, India
2 Department of Food Nutrition and Dietetics, Milagres College, Hamapankatta Mangalore, India
3 Department of Chemistry, Karmaveer Bhaurao Patil College, Vashi, India
4 Department of Chemistry, Veer Wajekar A.S.C. College, Phunde, India
Abstract
In the last few decades, nanoparticles have been analyzed and employed in various industrial applications. Because of numerous features such as antifungal, photochemical, high catalytic, and antibacterial activities, cobalt oxide nanoparticles have attracted a lot of attention because of high incidence of dangerous compounds and the harsh environments utilized in chemical and physical procedures of synthesis of different nanoparticles. Green Nanoparticles manufacturing approaches have been developed by employing plants, fungus, bacteria, and algae. There is a lot of research exploring numerous green production techniques in regulation to present confirmation of cobalt oxide nanoparticles of applications, biological applications, and non-hazardous effects. As an outcome, we have to gather pertinent review articles from inferior sources. Compared with chemical and physical preparation methods, the green synthesis route appears to be safer and extra environmentally forthcoming for the green preparation of the nanoparticles. However, its biomedical applications in this industry are daily in various procedures such as bio-imaging, biosensors, medication administration, and gene delivery. Moreover, cobalt oxide nanoparticles can operate as smart weapons against many drug-resistant microbes and are a talented antibiotic substitution due to their toxicity facial appearance.
Graphical Abstract
Keywords
Main Subjects
- Introduction
Nanotechnology has made significant progress in recent years and is one of the most rapidly growing concepts in science and technology. In a wide range of sectors, nanomaterials which have unique physicochemical properties, can build innovative systems, structures, nanoplatforms, and devices [1-4]. Nanomaterials have excellent catalytic reactivity, nonlinear optical performance, chemical stability, thermal conductivity, and a large surface area-to-volume ratio [5-6]. Because of this characteristic, several academics have looked for new ways to prepare. Traditional techniques involving physical and chemical processes require different hazardous compounds such as defensive agents to maintain stability, resulting in environmental toxicity. Plant extract-mediated nanoparticle biogenesis is cost-effective and supplies natural cap agents in the form of proteins. Therefore, green technology based on plants is gaining appeal as an ecologically acceptable, non-toxic, and safe option [7]. Plant extraction is used to normalize chemical toxicity in the environment from side to side biological synthesis of various metal oxide and metal nanoparticles, which is a marginal technique to regulate the chemical synthesis and allows for a different shape and size of nanoparticles with careful synthesis [8]. Biomedicals continue to be a hotbed of research into biodegradable, functionalized, and biocompatible nanomaterials. Paramagnetic nanoparticles, quantum dots (QDs), and carbon nanotubes (CNTs) nanoshells [9-11] have all been researched extensively, among other biological applications [12-15]. Cobalt oxide nanoparticles are present in various nanostructures and possess unique properties such as semi-conductivity, piezoelectricity, and opticality [16, 17]. As a result, cobalt oxide nanoparticle-based nanomaterials are being evaluated for various applications, including nanosensors, energy storage, cosmetics, nanoelectronic devices, and nano-optical devices [18-25]. Biodegradability and low toxicity are two of the essential characteristics of cobalt oxide nanoparticles. For adults, Co2+ is a vital trace element involved in many processes. Under strong basic and acidic conditions, cobalt oxide nanoparticles dissolved slowly. The release of CO2+ ions from soluble cobalt oxide nanoparticles has been shown to stress cells and cause harm to a variety of species [26]. In biological applications, the properties of cobalt oxide nanoparticles are becoming increasingly significant [27]. The solubility of cobalt oxide nanoparticles is linked to their detrimental effects. The nanoparticles of extracellular cobalt oxide nanoparticles are dissolved, gradually elevating intracellular [Co2+] levels. The mechanism of enhanced intracellular [Co2+] and cobalt oxide nanoparticles dissolution in the media is unclear [28]. The current state of cobalt oxide nanoparticles for biomedical applications, their green manufacturing nature, and their harmful impact will all be highlighted in this paper. Inorganic and organic nanoparticles are two types of nanoparticles based on their composition. Organic nanoparticles (fullerenes) are an example of organic nanoparticles. In contrast, inorganic nanoparticles include noble metal nanoparticles (gold and silver), semiconductor nanoparticles, magnetic nanoparticles, and (such as cobalt oxide nanoparticles and titanium dioxide) [29]. Nanoparticles can be categorized based on their origin, size, and structural composition. Natural and synthetic nanomaterials are classified into two groups based on their origin [30]. Based on their dimensions, nanomaterials are classed as three-dimensional (3D), zero-dimensional (0D), one-dimensional (1D), two-dimensional (2D), and One-dimensional nanomaterials have just one nano dimension outside the nanoscale range; nanomaterials of two-dimensional have two nano dimensions outside the nanometre range, and nanomaterials of three-dimensional have all nano dimensions outside the nanometre range. Immensity materials are included in discrete nanometre-scale blocks (1–100 nm) [31]. Nanomaterials are amalgamated materials or nanodispersions based on their structural arrangement and shape. Dendrimers are nanometer-scale macromolecules with a lot of branching. Metal which comprises metal oxides nanosilver, nanogold like titanium dioxide, and closely packed semiconductors like quantum dots, is the major component of these particles in metal-based materials. Carbon-based nanomaterials have morphologies such as tubes, hollow spheres, and ellipsoids. Nanotubes are cylindrical carbon nanomaterials, whereas fullerenes are spherical and ellipsoidal carbon nanomaterials [32].
This synthesis work is of high importance in determining different methods for the synthesis of cobalt oxide nanoparticles. Furthermore, it also gives biomedical applications along with the toxicity data of the cobalt oxide nanoparticles
2. Nanoparticles synthesis methods
Two approaches are advocated for nanoparticle biogenesis: bottom-up and top-down [30]. The most important process in a bottom-up approach is oxidation/reduction. Nowadays, nanoparticle synthesis is a prevalent issue in science, with researchers striving for an ecologically friendly process and green materials [31]. The synthesis solvent intermediate, the environmentally kind reducing mediator, and the non-toxic material for nanoparticle stability are the three grave steps in developing nanoparticles that should be evaluated from the aspect of environment-friendly approach chemistry. Thus, the majority of chemical and physical reactions described far employ organic solvents. The water-repelling capacity of various used capping agents is the primary cause [32]. Green chemistry principles are compatible with bio-organism synthesis, including: (i) an ecologically friendly method, (ii) the reduction of the used agent, and (iii) the reaction rounding agent. The synthesis of metal oxide inorganic nanoparticles by using materials biological has piqued curiosity due to their unique properties (optical, electrical, chemical, etc.) [33, 34]. Piezoelectric devices, fuel cells, microelectronic circuits, sensors, surface passivation coatings, and corrosion catalysts are just a few applications for metal oxides. Metal oxides have also been used in the environment as absorbers of pollution. Cobalt oxide nanoparticles are semiconducting metal oxide type that can display unusual chemical features in nanotechnology due to their tiny mass and high limit density, especially in biomedical systems, optics, and electronics [35–42]. Cobalt oxide NP stand out among the metal oxides because of their refreshing properties [43] (such as 3.3 vertical bandwidth at room temperature and excitation power of 60 meV) [44], high catalytic activity, anti-inflammatory, wound healing, and UV filters [45–49]. Several authors have reported cobalt oxide nanoparticles as biosensors for cholesterol, enzyme biochemistry, and other bio-sensing applications [50-52].

Figure 1. General synthesis of nanoparticles
Cobalt oxide NP is non-hygroscopic, non-toxic, and commonly found in organic, polar, crystalline material that has aroused interest in organic transformation, sensors, visible conductors, and surface acoustic wave devices [53-57]. Transparent electronics, UV light emitters, chemical sensors, spin electronics, personal care products, catalysts, adhesives, and paint are cobalt oxide NP. Due to its unique properties, cobalt oxide NP is used in light-emitting coatings, transparent electrodes in solar cells, UV light emitters, diode lasers, varistors, piezoelectric devices, spin electronics, electronics [58-60], and acoustic wave propagators [61] as an antibacterial agent [62] as a photonic material [63] and as a chevalier gas sensor Chevalier Biomolecules in the extract plant act as active capping agents, which assist in the production of nanoparticles. Capping agents stabilize NPs through various processes, including electrostatic stability, steric stabilization, hydration stabilization, and Vander Waals force stabilization. The NP stability is essential in its operation and implementation [64]. When applied to biodegradable polymeric metrics and cobalt oxide nanoparticles NPs, cobalt oxide nanoparticles in the food storage and packaging industry have improved food quality and packaging primarily through antimicrobial ion release, integrity destruction of viral cells, and ROS formation due to light rays [65]. Elmer and White investigated the pesticide capabilities of synthesized cobalt oxide nanoparticles by spraying them on tomato and eggplant plants, finding that it decreased illness by 28% compared with the control [66]. Plant extraction uses in vitro techniques to reduce cobalt salts (Cobalt nitrate, sulphate, chloride, etc.) and provide control over the size and condition of nanoparticles. The saponins, tannins, starch, polypeptides, terpenoids, flavonoids, and phenolics are the primary and secondary metabolites found in plants that act as reducing agents and capping. Plant metabolites are extracted by using mild solvents such as water, ethanol, and methanol which are allowed to react with a solution of cobalt salt in various conditions to obtain high yields [67, 68]. Several methods have been used to make nanoparticles of cobalt oxide NPs by using various sources such as bacteria, fungi, algae, plants, etc., due to the growing popularity of green methods. A table has been created to summarize the searches performed in this field, plant-mediated cobalt oxide nanoparticles NP biosynthesis, biological nanoparticles synthesis, is an effective mechanism for physical and chemical processes of nanoparticle creation. Several studies have focused on the nanoparticle synthesis of raw nanoparticle production of iron and oxide. Plant-based nanoparticle production provides a fast, inexpensive, environmentally friendly, and safe way for humans. With cobalt nitrate hex hydrate as a precursor, vitex and gundo plant extracts were recruited to form cobalt oxide nanoparticles, antimicrobial activity of biosynthesized cobalt oxide nanoparticles NPs against E. coli and S. aureus bacteria [69], and trifolium pratense was recruited [70] to form cobalt oxide nanoparticles. A hexagonal form of cobalt oxide nanoparticles were obtained with a 60–80 nm wavelength. In addition, the author tested the effectiveness of antidandruff, antimicrobial, and antiarthritic biosynthesized cobalt oxide nanoparticles [71-80]. Cobalt oxide nanoparticles produced from the extraction of green tea leaf and tested their behavioural potential in super-capacitors. Cobalt oxide nanoparticles are many inactive nanoparticles primarily used to treat urinary tract infections. Generated cobalt oxide nanoparticles from passiflora caerulea leaf extract have also been tested against pathogenic culture found in urinary tract infection f the patient. The findings revealed that the produced cobalt oxide nanoparticles had antibacterial properties against urinary tract infections, and investigated the cheap, non-toxic raw production of cobalt oxide nanoparticles by using Camellia sinensis extract to examine the efficacy of cobalt oxide nanoparticles as a photocatalyst to destroy various natural dyes such as methylene blue and methyl orange and their antioxidant activities by using Eucalyptus globulus. For the first time, they demonstrated a composite of mono-phase crystalline cobalt oxide nanoparticles with a diameter of 15.8 nm in a green and acceptable font in the area by using A-extraction, betulina as an effective oxidizing/reducing agent. Cobalt nitrate and Aloe vera leaf extract made cobalt oxide nanoparticles stable and circular. UV-Vis spectrophotometer analysis, FTIR, photoluminescence, XRD, SEM, and TEM were used to investigate different aspects of cobalt oxide nanoparticles [81].
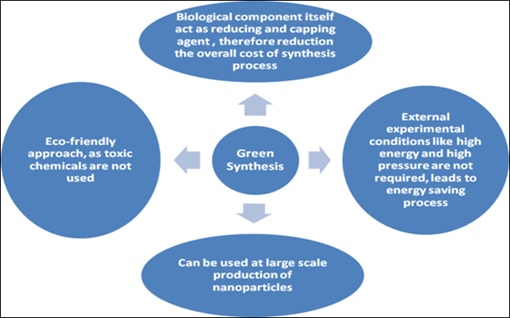
Figure 2. Green Method of synthesis [82]
3. Characterization of cobalt oxide nanoparticles (Co NPs)
EDAX (power dispersion evaluation of X-ray), AFM (atomic force microscopy), XPS (X-ray photoelectron microscopy), ATR (attenuated standard mirrored photograph), UV-DRS (UV-seen diffuse reflectance spectroscopy), XRD (X-ray diffract meter), TEM (transmission electron microscopy), and TG-DTA (thermogravimetric differential thermal evaluation) were used to investigate different aspects of cobalt oxide nanoparticles. Flora in the Lamiaceae own family, such as Vitex Segundo [83-87], Plectranthusamboinicus, and Anisochilus carnosus [88] had been significantly studied for the NP production in diverse paperwork, such as hexagonal, rod-shaped with agglomerates, quasispherical, round, and numerous diameters. The outcomes show that increasing the quantity of a plant extract lowers the generated NPs scale [88-90]. The size variety is viewed and compared with the use of diverse strategies, including TEM, XRD, and FESEM [91-94] which had a narrower variety of values, albeit SEM and EDAX produced similar findings that differed from the XRD effects. The XRD examination indicated that the NPs synthesis from each flower and the leaf of Vitex negundo had a comparable size of 38.17 nm. Cobalt oxide nanoparticles NPs are mechanically synthesized by using Azadirachta indica leaves from the Meliaceae own family [95-97]. Every experiment produced a comparable size variety of NPs, as evidenced by nanobuds, hexagonal disc shape, and round shape in TEM and XRD. The research suggested that amine, carboxylic acid, carbonate moieties, alcohol, alkane, and amide play a function inside the NPs manufacturing supported through FTIR research. Aloe vera leaf peel and fresh leaf extract belong to the Liliaceae circle of relatives [98-100]. Agglomerate formation changes were seen within the NP synthesis isolated from Moringa oleifera, Calotropis gigantea, Plectranthusamboinicus, Agathosmabetulina, Nephelium lappaceum, and Pongamia pinnata. UV-Vis spectrophotometry is utilized to validate NP synthesis, and crystal NPs are made via centrifuging the combination and drying the pellet in a hot air oven.
4. Biomedical applications of cobalt oxide nanoparticles
In biomedicine, cobalt oxide nanoparticles are used to deliver the drug. Their applications in drug delivery come from two main factors. First, nanoparticles may pass through tiny capillaries and are absorbed by cells due to their small size, allowing for the accumulation of effective drugs in the target areas. Second, organic matter for nanoparticle production enables long-term drug release in the target area [101]. The antibiotic metronidazole benzoate and its ointment were used to study the effect of cobalt oxide nanoparticles produced on drug release. According to the findings, including cobalt oxide nanoparticles in medicine has a significant impact on the membrane of biology.

Figure 3. Applications of cobalt oxide nanoparticles
5. Bio-imaging of cobalt oxide nanoparticles particles
Because it is inexpensive and straightforward, fluorescence imaging is commonly used in pre-clinical research [102-104]. Since cobalt oxide nanoparticles have significant blue emissions and near-UV UV, with green luminescence corresponding to O2 spaces, it has been used for cellular imaging in previous studies [105]. Small cytotoxic transferrin-conjugated green fluorescent cobalt oxide nanoparticles were used for cancer cell imaging [106]. Adding relevant components to cobalt oxide nanoparticles can affect their physical properties [107]. Cobalt oxide nanoparticles were contaminated with cations such as Co, Cu, or Ni, and then concentrated in aqueous colloidal solutions before being used in various cells to test cell images [108]. These tiny cobalt oxide nanoparticles have the potential to reach the cell.
Hetero-structural Co/Au Nanocomposites were developed and tested for biocompatibility and optical properties [109]. Au NCs thrive near the nanorod's surface or at the end of the cobalt oxide nanoparticles nanorods. Cobalt oxide nanoparticles nanorods linked to anti-epidermal growth factor antibodies were used to age in vitro cancer cells in recent activity [110]. QDs are generally considered nanoparticles for optical imaging because of their many attractive properties [111]. Cobalt oxide nanoparticles QDs were detected in the cytoplasm while used for in vitro cell imaging, showing continuous light under UV light without significant cytotoxicity. A previous study using mice with intravenous and intradermal injections looked at similar QDs [112]. Each method has its advantages and disadvantages [113]. Nanomatadium can be made to work to be seen by using various imaging methods, leading to synergistic processes. When paired with smaller molecules, nanomaterials are better at multimodality thinking because their larger space allows more functional sites and makes it easier to design multimodal acquisitions. In one study [114], Gd-doped cobalt oxide nanoparticles QDs (with a diameter of less than 6 nm) were helpful in MRI and optical imaging. Singh has published Fe3O4-cobalt oxide nanoparticles and core-shell magnetic QDs for cancer imaging and treatment. On the other hand, single-photon emission computed tomography (SPECT) [115] and radionuclide-based thinking methods, such as PET [116] are more clinically significant than optical imaging. The PET and SPECT pathways are complex and multifaceted; with no restrictions on tissue penetrate.

Figure 4. Applications of cobalt oxide nanoparticles [117]
6. Drug delivery of cobalt oxide nanoparticles
Due to their variable surface chemistry, extensive surface location, and picture poisonous impact, among different things, cobalt oxide nanoparticles nanomaterials are versatile nanoplatforms not handiest in bio-imaging, but additionally in drug shipping packages. However, cobalt oxide nanoparticles were hazardous to cancer cells [118, 119] and bacteria and leukemic T cells [120] in vitro studies. The intrinsic blue fluorescence of cobalt oxide nanoparticles QDs became smeared with folate conjugated chitosan thru electrostatic interplay. It may be loaded with 75% performance with doxorubicin (a commonly) used chemotherapeutic medication) [121]. DOX was recommended to be entrapped by using hydrogen bonding and cooperation with the cobalt oxide nanoparticles' QD surface. While due to the hydrophilicity and fees, the aqueous stability of the cobalt oxide nanoparticles QDs enriched the outer chitosan layer. It is estimated that DOX is rapidly elicited at a healthy pH of 7.4, which should be considered in vivo and in vitro studies. One of the significant challenges of dendritic migration (DC)-based immunotherapy for most cancer types is developing a mechanism that can precisely deliver the correct antigen to DCs [122]. Nanomaterials are difficult to use for this application because of their large surface area. Fe3O4 Cobalt oxide nanoparticles. Core-shell nanoparticles with an average diameter of 16 nm have been used to transport carcinoembryonic antigens to DCs [123].
7. Gene delivery of cobalt oxide nanoparticles
The most difficult element is developing safe gene vectors that could save DNA from degradation while contemplating excessive-performance cell absorption of DNA. Cobalt oxide nanoparticles nanomaterials, which have confirmed promise in numerous studies, are among many nanomaterials utilized to investigate gene therapy programs and gene delivery. Cobalt oxide nanoparticles nanostructures, which resemble a 3-dimensional tetrapod, had been used to transport pEGFPN1 DNA (which carries the inexperienced fluorescent protein gene) to A375 human cancer cells [124]. Electrostatic interactions connected the plasmid DNA (plasmid DNA) to cobalt oxide nanoparticles nanostructures, and three needle-shaped legs favoured internalization of the hints for gene shipping into cells. Lack of cytotoxicity was located, which turned into attributed to the three-dimensional geometry. The floor coating of nanoparticles is important for powerful gene shipping. Consistent with a take a look at [125], cobalt oxide nanoparticles QDs have been stacked with the use of definitely charged poly(2-(dimethylamino)ethyl methacrylate) (PDMAEMA) polymers for condensing DNA for gene switch. The polymer-coated cobalt oxide nanoparticles QDs fluoresced at 570 nm with a massive fraction of an awful lot, much less than 20%, condensing massive pDNA, including the luciferase reporter gene. In keeping with the researchers, COS-7 cells may be transfected efficaciously with pDNA that transmits cobalt oxide nanoparticles QDs with much less cytotoxicity and cytotoxicity cobalt oxide nanoparticles QDs changed into dramatically reduced whilst PDMAEMA end up used because the gene vector. The inclusion of negatively charged polymethacrylate in the QDs helped stabilize prices, lowering cytotoxicity.

Figure 5. Nano particles in drug discovery [126]
8. Biosensors based on cobalt oxide nanoparticles
Biosensors are commonly used in the food industry, environmental tracking, fitness care, and chemical and biological analysis. Electrochemical, photometric, piezoelectric, and calorimetric biosensors are examples of biosensors labeled with discovery concepts [127]. Nanomatadium, alone or in aggregate with natural compounds, draws developing hobby of their potential to offer an excellent platform for developing particularly efficient biosensors [128] along with antibodies, enzymes, and other proteins. They can also allow direct electron drift between electrodes and active areas of biomolecules. This component of semiconducting structures, cobalt oxide nanoparticles, also has bio-sensing, robust advertising electricity, high electric point, and extreme performance. Catalytic (IEP; nine. five) makes them appropriate for incorporating electrostatic adsorption of specific proteins together with antibodies and enzymes with low IEPs [129].
9. Toxicity studies of cobalt oxide nanoparticles
Cobalt oxide nanoparticles and TiO2 NPs were used. The ionic form of cobalt, which is highly toxic, is less harmful than cobalt oxide nanoparticles (NPs). The toxicity of cobalt oxide nanoparticles produced raw in zebrafish embryos [130, 131]. The author made cobalt oxide nanoparticles (NPs) from Amaranthus caudatus leaf extract. In addition, cobalt oxide nanoparticles had been found to have significant antibacterial activity toward Staphylococcus epidermidis and Enterobacter aerogenes. Toxicity studies determined that cobalt oxide nanoparticles at a dose of 10 mg/ml had no sizeable impact on zebra fatal survival and incapacity. In a 90-day toxicity look, Sprague Dawley mice were given a hundred nm cobalt oxide nanoparticles had a better charge [132] and were better charged, determining the risk level and identifying target organs. At concentrations above 125 mg/kg, both sexes show significant toxicity.
10. Conclusions
According to the research on green cobalt oxide nanoparticles, NP production is much safer and ecologically friendly than physical and chemical methods. Because of its numerous properties, capacities, benefits, and application to humans, cobalt oxide nanoparticles are one of the most essential and adaptable materials. In the arrival of regulated length and shape nanoparticles, raw materials operate as stabilizing and reducing agents. In plants, cobalt oxide nanoparticles sustain moderate growth and crop yield. At the same time, despite the inadequate yield of a critical crop, demand for meals continues to rise. Therefore, metallic oxide nanoparticles should be marketed for agricultural sustainability over a long period. Bio-imaging, medication management, biosensors, and gene transfer are just a few of the organic uses in this field that are rapidly growing.
Furthermore, cobalt oxide nanoparticles can be cunning weapons against numerous drug-resistant pathogens and a capable antibiotic potential because of their toxicity. This overview should make it easy to research breakthrough methodological and scientific connections in this area. In the meanwhile, health-related concerns will be handled.
Acknowledgment
The authors would like to acknowledge the management and principal of supports.
Orcid:
Rupali C Shete
https://www.orcid.org/0000-0003-1694-2709
Citation: R.C. Shete*, P.R. Fernandes, B.R. Borhade, A.A. Pawar, M.C. Sonawane, N.S. Warude. Review of Cobalt Oxide Nanoparticles: Green Synthesis, Biomedical Applications, and Toxicity Studies. J. Chem. Rev., 2022, 4(4), 331-345.


