Document Type : Short Review Article
Authors
1 Department of Chemistry, Shahreza Branch, Islamic Azad University, 311-86145, Shahreza, Isfahan, Iran
2 Department of Chemistry, Faculty of Sciences, University of Hormozgan, Bandar Abbas 71961, Iran
Abstract
Organic polymers are one of the most abundant groups of materials, and plastics, rubbers, fibers, adhesives, and coatings are examples of the attractive world of polymer chemistry. Natural polymers usually have a more complex structure than synthetic polymers, so the study of synthetic polymers is particularly important in terms of building simplicity. In this work, the properties and applications of polymers are discussed. Likewise, three important types of polymer compounds including polyurethanes, polyesters, and polyimides were investigated.
Graphical Abstract
Keywords
Main Subjects
- Introduction
Macromolecules are formed by the bonding small hydrocarbons together during the polymerization process. Polymerization reactions take place in two ways: growth-step polymerization and incremental polymerization. In the first method, polymerization takes place through condensation reaction between multifactor molecules, and in the second method, monomeric molecules are added to the growing chain over time and will not have a by-product reaction [1].
Organic polymers are one of the most abundant groups of materials and their wide applications as plastics, rubbers, fibers, supercapacitor, batteries, pharmacology, adhesives, and coatings are some examples of the attractive world of polymer chemistry. Polymers are classified on several bases. One type of classification is based on the polymers origin. Accordingly, polymer is either natural such as wax, wool, or synthetic rubber such as nylon or deformed such as cellulose nitrate, cellulose acetate, etc. Natural polymers usually have a more complex structure than synthetic polymers, thus the study of synthetic polymers is particularly important in terms of building simplicity. Another type of classification is based on the shape of the polymer, meaning that monomers react with each other in a process called polymerization to produce polymers in various forms. Based on these shapes, polymers can be divided into linear, railing, comb star, and three-dimensional. Polymers are also divided into two categories based on the type of monomers involved in the reaction: Homopolymers and copolymers. Homopolymers are polymers whose constituent monomers are of the one type. Copolymers are polymers made from more than one type of monomer. In another type of classification, polymers are classified according to the type of polymerization reaction.Polymer reactions were initially divided in 1929 by Carroters into incremental polymerization and condensation polymerization. This classification was based on the polymer’s composition and structure, and then, in 1950, this classification was modified based on the polymerization reaction mechanism by Mark and was divided into two categories: stepwise and chain polymerization [2-6].
- Investigation of properties of polymers
2.1 Thermal resistance of polymers
Stability of a polymeric material is the ability of a polymer to withstand a certain temperature and time without reducing or changing the polymer properties. Thermal stability of polymers is one of the important issues that has been considered by researchers in the last three decades. Therefore, polymers are exposed to various thermal agents, oxidizers, solvents, etc. during their application life, and their stability against these forces and destructive factors can be determined by measuring a number of mechanical properties and various tests. Polymers are subject to physical changes (reversible) or chemical changes (irreversible). Physical changes typically include changes in glass transition temperature, melting, crystallization, and polymer morphology indicating the thermothermal state of the material. The degradation action of these polymers depends on both heat and time, and the stability of the polymer is determined by the rate at which it decomposes at a given temperature [7].
There are four ways to improve thermal resistance of a polymer: (i) Increased crystallinity by rigid groups. (ii) Increasing cross-linking and networking. (iii) Add aromatic groups that increase inter-chain interactions. (iv) Addition of heat-resistant groups to the main chain such as triazines, phthalimides, and heterocycles such as phosphazine, oxazolidone, and some carboxylic acid derivatives such as imide, and amides and useful compounds such as ether and sulfon [8-13]. In 2012, Mansouri et al. in a study entitled: “Synthesis of heat resistant polyamides with hanging groups of 1,3,4-oxadiazoleby direct polymerization using 2-(5-(3,5-diaminophenyl)-1,3,4-oxadiazole-2-yl)pyridine (1) and aliphatic and aromatic carboxylic acid (Scheme 1) in ionic liquids as solvent concluded that polyamide with high heat resistance, low water absorption, and the highest molecular weight in Solvent 1-butyl-3-methyl imidazolium bromide was obtained [14].

Scheme 1. Synthesis of polyamide with oxadiazole hanging group
2.2 Solubility of polymers
An important part of identifying polymers is examining their solubility. Often for polymers that are unstable in the molten state, the method of industrial fabrication using their solution. The dissolution of polymers is different from the dissolution of low molecular weight materials, in polymers, dissolution consists of two steps. First, the solvent penetrates between the polymer filaments and the so-called swollen polymer, which is called a gel, swells. Likewise, in the next step, the gel loses its state and the strands become a real solution [15]. Various methods have been proposed to increase the solubility of polymers, including the introduction of flexible groups in the main chains such as groups such as sulfone (SO2), ether groups (-O-), and noted alkyl and sulfide groups. The aryl ether group increases the solubility and causes a slight decrease in thermal stability. The hexafluoroisopropylidene group increases the polymer solubility and modifies its properties including thermal stability, oxidation, and resistance to flame and environment [16].
In 2003, Grath et al. investigated the physical and chemical properties of a sulfonated copolymer (arylene-ether-sulfone) (6) according to (Scheme 2). The resulting copolymers had good solubility due to the presence of sulfone and ether groups in the main chain. In addition to increase the strength due to their flexibility, these groups increase the solubility of the polymer. In 2021, there was a report of the use of polysulfide polymers were synthesized using organic monomer (ethylene dichloride) and sodium-based aqueous monomers via interfacial polymerization. The results showed that along with increasing sulfur in the structure of polymers, solubility were increased whereas it decreased the hardness, melting point, and glass transition temperature of the obtained polymers [17, 18].

Scheme 2. The structure of the copolymer (arylene-ether-sulfone) is sulfonated
2.3. Degradability of polymers
The stability of polymers is a very important factor in the properties of polymers, and has led to many studies on the polymers degradability [19]. Thermal gravimetric analysis is one of the methods to study the degradation of polymers. In this method, weight loss is measured as a function of temperature or time at a specific temperature. The graph shows the changes in temperature or time versus weight loss percentage, which will be valuable in identifying heat-resistant polymers [20].
2.4. Optical activity in polymers
Most natural macromolecules, such as nucleic acids, proteins and chiral polysaccharides, are optically active. In the study of the chirality of nearly 800 drugs derived from natural sources, only 2% are racemic, 1% non-chiral, and the rest are optically active. Chirality for the polymer is a key structural factor that is responsible for some complex properties such as molecular detection and catalytic activity [21, 22].
- Polyurethanes
Polyurethanes are a unique class of polymers been used in a variety of forms [23] and they have tensile strength, high strength, and other excellent mechanical properties and are resistant to radiation, heat, oils, acids, and bases. By selecting the appropriate catalyst, both the speed and the path of polyurethane formation process can be controlled. Commercially used catalysts in the process of preparing polyurethanes, tertiary amines, and organic compounds are tin [24, 25]. The first polyurethane in 1937 reported by Bayer et al. Thermoplastic polyurethanes are the result of a single-step or multi-step reaction that results in the expansion of the chain and the formation of a high molecular weight and soluble polymer. Polyurethane elastomers have a higher load bearing capacity under equal conditions in terms of elasticity and stiffness compared to other elastomers. This advantage means that they can be used for similar applications at a lower weight than other elastomers. The electrical properties of polyurethane elastomers are good and they are similar to phenolic resins. Polyurethane types of hard elastomers have better electrical properties than their foam type. Thermoplastic polyurethanes for elastomers are made on the basis of polyurethane. Polyethers have a lower coefficient of elasticity than polyesters. The amount of tensile strength in both types (polyester and polyether) is the same. The rate of longitudinal increase of polyether type is more than polyester of which its specific gravity is less than that of polyester. The flexibility and abrasion resistance of polyurethane is higher than that of polyester [26-31].
3.1. Basics of chemistry and technology of polyurethanes
Polyurethanes are prepared as a reaction product of isocyanates and polyhydroxy substances. It is obvious that reactive substances can have two or more active agents, in which case a network structure with more transverse connections is created. Two different types of polyol, including polyester and polyether, are widely used in the preparation of polyurethanes; the type of polyol selected is very effective on the final properties of the obtained product. Polyesters usually provide better mechanical properties and polyethers provide water resistance in polyurethanes, polyesters have better thermal stability compared with polyethers, and their resistance to oxidation is relatively good [32, 33].
Isocyanates were discovered by Wertz in 1849, and from 1937 onwards, their use in the production of polyurethanes began. There are two types of diisocyanates used to produce polyurethane: aromatic and aliphatic diisocyanates. Aromatic isocyanates in the hard part of polymer chain make the chain stiffer and increase the melting point of the polymer. Aromatic diisocyanates and polymers made of them are somewhat unstable against light and change color over time. However, polyurethane made from aliphatic isocyanates, in addition to be more stable against light, against decomposition being in water (hydrolyzing) and destruction due to heat also show a lot of resistance. Unfortunately, along with this property improvement, the mechanical properties of polyurethane prepared from aliphatic diisocyanate decrease. Aromatic diisocyanates are more reactive than aliphatic or cycloaliphatic ones [34-38].
The commercial method of producing isocyanates (11) is indicated in (Scheme 3) that the reaction of amines (9) with phosgene (10) is under the special conditions [39].
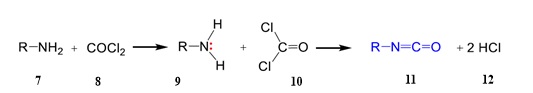
Scheme 3. Method of producing isocyanates
As shown in (Scheme 4), the isocyanate group (13, 14, 15) can have different resonance structures according to its electronic structure. The resonance balance below shows that the electron density is higher on the oxygen and nitrogen atoms and less on the carbon atom. In addition, this issue causes oxygen and nitrogen atoms to have the highest negative charge and carbon atom to have the highest positive charge. The group attached to NCO can increase or decrease the isocyanate activity, depending on whether it is electron withdrawing or electron donating. For this reason, aromatic isocyanates are more active than aliphatic isocyanates.

Scheme 4. Resonance of isocyanate group
Reaction with alcohols, amines, water, carboxylic acids, phenols, amides, ureas, and urethanes, as well as polymerization, dimerization, trimerization, Friedel Crofts, and Grignard reaction, the reaction of unsaturated compounds, and the reaction with halogens are among the reactions of the isocyanate group with other organic compounds [40-45].
3.2. Application of polyurethanes
Polyurethane is used in elastic coatings in the textile industry, adhesives for sheets, and thin films. Polyurethanes are used in paint and coating industries due to their good abrasion resistance. Flexible polyurethane foams are used to make sleeping mats, supports, and car seats. Hard foams are used to insulate freezers, refrigerators, and roofs. One of the most important applications of polyurethane is its use in the manufacture of sports equipment and sports aids. Polyurethane can act to protect air and soil from contamination. In the mining industry, polyurethane injection is used as a very good absorber of pollution, and also low-density polyurethane injection and fast curing are used to strengthen the mine wall [46-48].
3.3. Relationship between properties and structure in polyurethanes
Polyurethanes are polymers in which hard sections of urethane are embedded in soft sections, typically polyether or polyester. The soft parts give the resulting polymer elasticity, while the hard parts increase the polymer hardness due to their intermolecular gravitational force. The presence of intermolecular attractions allows the polymer to retain its physical and mechanical properties at high temperatures. Hard parts in batch polyurethane affect their mechanical properties such as modulus, hardness, and tear strength and determine the highest application temperature of these materials, while soft parts have an elastic role. They are responsible for the chain and affect the properties of the polymer at low temperatures [49-51].
3.4. Synthesis of polyurethanes
In the synthesis of polyurethanes, the word block ratio, NCO:OH ratio is very common, this parameter controls the reaction and is related to the stoichiometric ratios of the material. Because the reaction efficiency between NCO and OH groups is high, by choosing the molar ratio of these two groups, the structure of urethane will be predictable [52]. Techniques commonly used in the urethanes synthesis include pre-polymer, semi-prepolymer, and single-step. In the first method, the reaction of a polyol with higher amounts of polyurethane diisocyanate is obtained. In the second method, part of the polyol is reacted with all the diisocyanates and the semi-pre-polymer is obtained, and then the mixture is mixed with the rest of the polyol to obtain the final polymer. The one-step method is the simultaneous mixing of poly-stoichiometric values, diisocyanate, and incremental chains. In this method, the control reaction is not arranged. Therefore, the structure of polyurethane will be random [53-56].
In 2022, Li et al. A green and facile method was proposed to fabricate waterborne polyurethane (WPU) by recycling waste polyester. It was found that WPU was successfully prepared by using bis(2-hydroxyethyl) terephthalate (BHET) as chain extender. Compared with polyurethane prepared by small molecular chain extender 1, 4-butanediol, mechanical strength, and thermal stability of polyurethane were remarkably enhanced by the BHET incorporation. Meanwhile, water absorption rate of polyurethane is reduced [57].
In 2005, Behniafar et al. synthesized a completely new aromatic polyurethane-imide (18) (Scheme 5), and the resulting product showed excellent solubility in polar solvents. Furthermore, increasing the thermal stability of homopolyurethane by thermal gravimetric analysis (TGA) proved that weight loss of 10% under nitrogen is done in the temperature range of 391-411 °C [58].

Scheme 5. Synthesis of polyurethane-aromatic imide with phthalimide group
In 2013, Erbay et al. prepared a diol containing the sulfone and ether groups using bisphenol sulfonate and ethylene carbonate and synthesized an acrylate polyurethane (29) (Scheme 6) [59].

Scheme 6. Acrylate polyurethane prepared by pre-polymer method
- Polyesters
4.1 History
Polyester was initially introduced as a polymer by Carrots. In fact, he discovered that fibers could be successfully obtained from a combination of alcohol and carboxylic acids. In 1939, a group of British scientists followed Carruthers research and in 1941 succeeded in producing the initial polyester fibers. The reason for choosing the name polyester is due to the presence of repeated connections of the ester group (-CO-O) in their main chain, it is a polar group that induces intramolecular and extramolecular chain interactions. These interactions appear to be responsible for various features, including low solubility, chain mobility, and melting. Therefore, research is needed to improve existing systems, to increase the desired applications and properties. For instance, the introduction of flexible groups such as esters, sulfones, and ether parts, which increase polymer solubility without much change in thermal resistance [60-62].
4.2. Properties and application of polyesters
Polyesters have a variety of properties including strength, resistance to most chemicals, corrosion resistance, abrasion resistant, easy to wash, stretch, and shrinkage resistance. Due to the good quality of polyester fibers and fabrics, these materials have various applications. Due high hardness and durability, polyester is often used as a garment and clothing. Its hydrophobic properties make it an ideal material to use in wet or damp environments, since polyester can be molded in any way, certain insulating properties can be created in it, one of which is to make hollow fibers. This process traps air inside the fibers, which is heated by the human body. This warm air stays inside the fibers and warms the body cold air. The second method is to use twisted fibers in fiber-filled products. Twists keep the air warm. Moreover, polyester is used in numerous industrial products, including carpets, filters, synthetic vessels, ropes, films, and fabrics for automobiles [63-68].
4.3. Synthesis of polyesters
Polyesters are obtained by the direct esterification of a diacid and a diol, and also by a self-compressing hydroxycarboxylic acid reaction. As polyesterization is an equilibrium reaction, like many other step polymer processes, water should be constantly removed until a high degree of conversion or high molecular weight is reached. Polymer temperature control is important to prevent adverse side reactions such as dehydration of diol [69, 70].
In 2021 Cai et al. prepared polyesters using catalyst-free conditions. Catalyst-free polyesters with high molecular weights were synthesized via a tandem mechanism involving anhydride formation and re-esterification using commercial primary diols and excess dicarboxylic acids that can form cyclic anhydrides. These methods can produce polyesters comparable to current commercial products while avoiding the negative effects caused by catalysts, which is very promising for the applications with high safety requirements [71].
In 2021, Wan et al. prepared green polyester and put forward a solution for the direct preparation of green copolyesters with versatile functionalities via the cascade polycondensation-coupling ring-opening polymerization) using a biobased cyclic diester ethylene brassylate as the monomer and functional diols/polyether diols as initiators. The combination of both polymerization processes into a system enables the facile synthesis of biodegradable polyesters with versatile functionalities, including alkane, alkene, alkyne, aromatic, halide, ether, and amide [72].
- Polyimides
5.1. Properties and applications of polyimides
Polyimides are high-performance polymers, which are most widely used in engineering polymers. Polyimides are used as high-temperature plastics, adhesives, dielectrics, light-resistant materials, and membrane materials for separating membranes. They are also used in aerospace, defense, electronics, composite, and optical fiber industries. Polyimides were initially synthesized and known in 1908. But in practical terms, they did not find commercial use until the late 1950. In 1950, DuPont introduced these polymers commercially, and since then, a large number of polymers have been synthesized. It should be noted that many companies are currently active in the field of preparing and producing these polymers [73-75].
High strength composites, thermally stable films, molding compounds, and adhesives can be prepared from polyimides. In addition to thermal stability, stability against oxidizing and hydrolytic substances and good mechanical properties are characteristics of polyimides. Aromatic polyimides are of interest due to their high resistance to oxidative degradation and heat stability. Increasing the number of conjugated aromatic rings increases the polymer hardness. The existence of a rigid structure and compression of polyimide chains increases the polymer crystallinity and increases the glass transition temperature [76].
Despite the excellent properties of polyimides, due to their high melting temperature and glass transition temperature, their use is limited because their processability becomes difficult. Therefore, various ways have been proposed to improve the properties of polyimides: (i) Addition of flexible groups (related to diamine or dianhydride) such as ether and carbonyl [77]. (ii) Preparation of some copolymers such as poly(ether-imide) (33) (Scheme 7) [78] and poly(ester-imide) (36) (Scheme 8) [79]. (iii) Addition of a bulky hanging group in the main chain, such as phenyl, benzoxazole, naphthalene groups, etc. which increases the space between the polymer chains, prevents imide and aromatic rings from being in the same plane, and reduces the ability to form charge transfer and bond complexes. Hydrogen moves between materials. They also increase thermal and oxidative stability [80].

Scheme 7. Synthesis of poly(ether-imide)

Scheme 8. Synthesis (ester-imide)
5.2. Synthesis of polyimides
(i) Polyimidization by condensation method, in which polyimides are prepared either from the reaction of diamine with dianhydride or tetracarboxylic acids, after passing through the amic acid stage, and then by losing water [81]. (ii) Additive polyimidization, which involves the polymerization of compounds with low molecular weight carrying imide groups inside the compound structure. These monomers have active groups and are subject to incremental polymerization due to heat and lead to the formation of network polymers [82].
- Conclusion
This work discusses and analyzes the uses and properties of polymers, polymers are very diverse compounds and have been used in various fields of industry, such as plastics, rubbers, fibers, adhesives, and coatings. These compounds demonstrated great potential in various fields of chemistry and industry. The unique properties of polymers are promising for further use of these compounds.
Acknowledgment
The authors would like to appreciate Dr. Majid Kolahdoozan for supporting this research study.
Orcids:
Omid Soleimani:
https://orcid.org/0000-0001-5986-7475
Hosein Mahmoodi Khaha:
https://orcid.org/0009-0006-1096-1499
Citation: H. Mahmoodi Khaha, O. Soleimani*, Properties and Applications of Polymers: A Mini Review. J. Chem. Rev., 2023, 5(2), 204-220.


