Document Type : Review Article
Authors
Department of Pharmaceutical Chemistry, St. Joseph’s College of Pharmacy, Cherthala, Kerala, India
Abstract
The fact that pyridine-based ring structures have a strong impact on pharmacological activity and have been used so frequently in the drug development process is largely responsible for the discovery of numerous broad range medicinal medicines. Pyridine is a fundamental heterocyclic chemical molecule with an identical conjugated six-electron system to benzene. With the rise in popularity of niacin in 1960, pyridine became an intriguing target. Pyridine and its derivatives are widely distributed in nature, where they serve a crucial role in heterocyclic chemistry and numerous uses in the field of medicine. Globally, 110 million individuals were expected to have diabetes in 1994, and 239 million were projected to have it by 2010. It was reported in 1998 that 135 million people had diabetes in 1995 and that 300 million people would have the condition by 2025. According to a later study, the number of persons with diabetes worldwide increased from 171 million in 2000 to 366 million by the year 2030. According to the International Diabetes Federation's most recent projection, there would be approximately 600 million people living with diabetes worldwide by 2035, up from the previous estimate of 382 million for 2030. Given that diabetes is a global pandemic, it is clear from all of these data that novel anti-diabetic medicine formulations are required. The anti-diabetic properties of various pyridine derivatives from the research articles published up to this point are the focus of this review study.
Graphical Abstract
Keywords
Main Subjects
- Introduction
Diabetes, a metabolic disorder with massive consequences, with estimated prevalence of 422 million patients[1, 2]. By 2030, the International Diabetes Federation projects that there will be 552 million diabetic patients worldwide. Age, ethnicity, smoking, obesity, and physical inactivity are the main risk factors for this increased risk of diabetes. Type 2 diabetes or non-insulin dependent diabetes affects 85–95% of people (T2DM) [3, 4]. T2DM is characterized by poor glycaemic control brought on by insulin resistance and insufficient pancreatic insulin secretion [5–10], which opens the door to irreversible complications [11, 12] as eye damage, heart and blood vessel disease, nerve damage, kidney damage, hearing impairment, etc. Less than one-third of T2DM patients achieve target glycaemic control even after various pharmacological medications are available because of issues with the metabolic syndrome. Studies have shown that diabetes increases the severity and mortality of COVID-19 patients and that diabetics are more likely than healthy individuals to develop the condition, as well as complications including ARDS and even fatalities [13]. For the treatment of patients with uncontrolled T2DM, it is necessary to discover new medications with novel mechanisms of action to supplement the current therapy. There has been a surge in interest in the creation of innovative anti-diabetic drugs ever since the ground breaking discovery of pyridine derivatives [14]. Moreover, specific synthetic pyridine derivatives have exhibited hopeful anti-diabetic activity.
Pyridine, on the other hand, is a fundamental organic heterocyclic molecule. Pyridine and its precursor, dihydropyridine, are among the most common structural units in the pharmaceutical industry [15]. They serve as important scaffolding in naturally occurring and biologically active compounds. A quick look at the US Food and Drug Administration (FDA) database reveals that roughly 14% and 4%, respectively, of the N-heterocyclic medications approved by the organisation contain pyridine and dihydropyridine [16] (Figure 1). The features of pyridines, such as their weak basicity, water solubility, chemical stability, capacity to form hydrogen bonds, and tiny molecular size, make them significant in the field of medicine. Synthetic chemists have recently focused on creating new analogues that use pyridine or dihydropyridine templates in their molecular design to research their mechanisms of action and find new medicinal leads. The sheer volume of publications demonstrates the importance of pyridine in chemical sciences and medical chemistry [17-19]. Extensively Pyridine and dihydropyridine containing drugs are mostly used as antimicrobial [20-22], antiviral [23, 24], anticancer [25-27], antioxidant [28, 29], antimycobacterial [30, 31], anti-diabetic, anti-malarial [32-34], antihypertensive [35, 36], and anti-inflammatory agents [37, 38]. Pyridine has got many derivatives [39] and each of those derivatives have a specific biological activity. The important pyridine containing drugs include niacin, nicotinamide, piroxicam, isoniazid, pyridostigmine, etc. [40]. We concentrate on the anti-diabetic properties of pyridine derivatives in this review.

Figure 1. The US Food and Drug Administration (FDA) database of the N-heterocyclic medications
Using DFT/B3LYP/ and TD-DFT, it is possible to successfully conduct a theoretical investigation of the geometric, electronic, and photophysical properties of modelled Bithiophene-bipyridine co-oligomers and their derivatives to elucidate their electronic structures, molecular reactivity descriptors, and predict their applicability in optoelectronic devices. Compared with bithiophene-bipyridine co-oligomers with substituents connected to pyridine, those with substituent attachment to thiophene have better optoelectronic characteristics.
- Pyridie and Its Derivatives as Anti-Diabetic Agents with Different Therapeutic Targets
2.1 α-Glucosidase inhibitors
An essential enzyme called α-glucosidase contributes significantly to the metabolism of carbohydrates by producing glucose and other sugars with lower molecular weights without having a direct impact on the production of insulin [41].
A considerable way to treat the type 2 DM is to inhibit the intestinal α-glucosidase [42]. Acarbose, voglibose, and miglitol are just a few of the α-glucosidase inhibitor-based medications that are readily available to treat type 2 diabetes. However, their side effects prevent them from being the ideal therapeutic agents for the treatment of DM [43]. Researchers were inspired to create and manufacture novel anti-diabetic chemicals, especially -glucosidase inhibitors (-GIs) due to the rising number of people with the disease. They prevent the small intestine from absorbing carbohydrates, which is how they work. Enzymes that transform complicated non-absorbable carbs into simple absorbable carbohydrates are competitively inhibited by them [44]. These enzymes include sucrase, maltase, glucoamylase, and isomaltase.
Based on an environmentally friendly process, Lingala et al. created a pharmaceutically intriguing type of functionalized dihydro-6H-chromeno[4,3-b]isoxazolo[4,5-e]pyridine heterocycles [45]. The anti-diabetic efficacy of the produced compounds against type 2 diabetes mellitus was assessed.
Therefore, compound (1) showed the highest level of -amylase activity inhibition (IC50=56.043 g/mL) (Scheme 1).

Scheme 1. Synthesis of compound (1)
Novel coumarin fused pyridine derivatives were successfully synthesised using a one-pot, three-component method by Mehdi et al. [46]. When the α-glucosidase inhibitory activity of the synthesised compounds was evaluated, all of them demonstrated remarkable -glucosidase inhibition of yeast in vitro, with IC50 values ranging from (101.0 2.0 to 227.3 1.4 M) when compared with the reference drug acarbose (IC50 = 750.0 1.5 M). The most effective substances were discovered to be (2), (3), and (4). According to a kinetic analysis of (2), it inhibited glucosidase in a competitive manner (Scheme 2).

Scheme 2. Synthesis of compounds (2), (3), and (4)
Different imidazo[1,2-a]pyridines attached to the carbamate moiety were devised and created by Saeedi et al. [47]. They were tested for their ability to inhibit α-glucosidase, with acarbose serving as the reference substance. When compared with acarbose, the reference medication, which has an IC50 of 750.0 M, 4-(3-(tert-butylamino)imidazo[1,2-a]pyridine-2-yl phenyl p-tolylcarbamate (5) was the most powerful molecule (IC50=75.6 M). Compound (5)'s kinetic investigation predicted a competitive inhibition (Scheme 3).

Scheme 3. Synthesis of compound (5)
In vitro testing was performed on 2-amino-4-aryl-6-(phenylthiol)pyridine-3,5-dicarbonitriles produced by Muhammad Ali et al. [48]. Their ability to inhibit α-glucosidase was assessed. The inhibitory potential of some analogues was significantly higher than that of the common acarbose (IC50=750 .10 M).
Interestingly, compound 6 had a thirteen-fold stronger inhibitory effect than the common acarbose (IC50=55.6 0.3 M). The most effective compound, (6), demonstrated a competitive type inhibitory action through kinetic tests (Scheme 4).

Scheme 4. Synthesis of compound (6)
Novel pyridine -2, 4, 6-tricarbohydrazide thiourea compounds were created by Tanzeel et al. [49] (Figure 2). These substances were tested for their ability to block the α-glucosidase enzyme. Compounds (7) (IC50 25.49 0.67 M), (8) (IC50 28.91 0.43 M), (9) (IC50 30.66 0.52 M), and (10) (IC50 35.01 0.45 M) provided superior inhibition against-glucosidase as compared with the reference molecule acarbose (IC50 38.22 0.12 M).

Figure 2. Pyridine-2,4,6-tricarbohydrazide thiourea compounds
The compound 2,6-diaryl-4-oxo-N,N′-di(pyridin-2-yl)cyclohexane-1,1-dicarboxamide was created and produced by Abdullah et al. [50]. The compounds with the highest α-glucosidase inhibitory activity were (11), (12), and (13) (Scheme 5).

Scheme 5. Synthesis of compounds (11), (12), and (13)
2.2 α-Amylase inhibitors
The enzyme α-amylase, which aids in the digestion of starch and glycogen, is regarded as a therapy option for conditions involving abnormal glucose absorption, such as obesity and diabetes [51]. α-amylase inhibitors inhibit the activity of salivary and pancreatic amylase in vitro and in vivo [52].
They can impair the growth and metabolism of animals [53] when ingested in large quantities, but may be useful in the management of diabetes or obesity.
Muhammad et al. created and produced 5-amino derivatives of nicotinic acid [54]. They were tested for their ability to inhibit the enzymes glucosidase and amylase. The synthesised compounds showed encouraging α-amylase activity, with values ranging from 12.17 0.14 to 37.33 0.02 g/mL SEM. Compounds 14, 15, 16, 17, 18, and 19 all showed notable α-amylase inhibitory actions, and their inhibitory potential was on par with that of acarbose (SEM: 10.980.03 g/mL) (Scheme 6).

Scheme 6. Synthesis of compounds (14-19)
The inhibitory activity of various pyridine derivatives was studied by Hyuk et al. [55] such as pyridoxine (20), pyridoxal (21), and pyridoxamine (22) (Figure 3). Pyridoxal significantly outperformed pyridoxine in terms of α-amylase inhibitory action (10.87 mg/mL of IC50 vs. 23.18 mg/mL of IC50). This suggests that pyridoxal can prevent pancreatic-amylase from hydrolyzing starch in the small intestine.
These findings suggest that the water-soluble vitamin pyridoxine and its derivatives can lower blood glucose levels by activating enzymes involved in absorption. Pyridoxal may therefore have the potential to be utilized as an additive in food to stop the progression of pre-diabetes to type 2 diabetes.
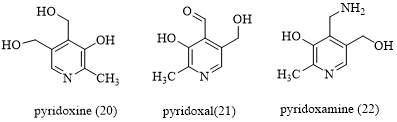
Figure 3. Pyridine derivatives
2.3 Dipeptidyl peptidase-4-inhibitor
Injurious microvascular and macrovascular problems are associated with T2DM, which affects more than 370 million people globally [56] such as retinopathy, nephropathy and coronary artery disease, foot damage, hearing impairment, Alzheimer’s disease, etc. Incretin hormones, glucose-dependent insulinotropic polypeptide (GIP) and glucagon-like peptide-1 (GLP-1) [57, 58], play a crucial role in the maintenance of normal glucose homeostasis. Both incretins can glucose-dependently invigorate insulin secretion and inhibit glucagon release, and also hold up gastric emptying and subdue appetite. In plasma, the active forms of these incretins are quickly metabolized by dipeptidyl peptidase-IV (DPP-4) [59-61]. Therefore, increasing both incretins plasma endogenous levels with DPP-4 inhibition can enhance T2DM patients' glycemic control. T2DM has been successfully treated with DPP-4 inhibitors as sitagliptin, linagliptin, alogliptin, and teneligliptin. DPP-4 inhibitors, either alone or in combination with other medications, can manage hyperglycemia with similar efficacy to sulfonylureas [62-64]. They exhibit good safety and tolerability characteristics, provide no inherent risk of inducing hypoglycemia, and have no effect on body weight [65]. However, these medicines continue to fall short of clinic and industry expectations, particularly for older T2DM patients. Here, we provide the results of a biological assessment of novel DPP-4 inhibitors called pyridine derivatives.
Several scaffolds made of imidazo[1,2-a] pyridine were created by Qing Li et al. [66] (Figure 4). They were assessed for potential DPP-4 inhibition action. Due to the structural changes made to compound 16's 2-benzene ring and pyridine moieties, compound 23 with a 2, 4-dichlorophenyl group at position 2 was identified as a powerful (IC50 = 0.13 M), selective (DPP-8/DPP-4=215 and DPP-9/DPP-4=192), and in vivo effective DPP-4 inhibitor. With the pyridine moiety of the imidazo[1,2-a] pyridine ring providing an extra -contact with Phe357 of DPP-4, additional molecular docking experiments showed that compound 23 might retain important binding characteristics of DPP-4.

Figure 4. Imidazo[1,2-a] pyridine
For the on-going development of a novel DPP-4 inhibitor for T2DM, compound 23 may be a viable lead.
Novartis reported altering the previously mentioned DPP-4 inhibitors in 1999 [67], L-amino acids containing a primary amine at their N-terminus in the P-2 site (Figure 5). The slow binding inhibitor (24) (NVP-DPP728) was chosen as a clinical development candidate for type 2 diabetes after pyridine collections were validated.

Figure 5. L-amino acids containing a primary amine
A series of pyridine-based DPP-4 inhibitors were revealed in a Takeda (Japan) (Figure 6) patent from 2005, and a second series of pyridyl acetic acid derivatives were revealed in 2006 [68] (26). With reference to position 5 on the ring, substituted 3-aminomethyl pyridines offer substantial flexibility without significantly changing the DPP-4 inhibitory action (25, 26, 27, and 28).

Figure 6. Pyridine-based DPP-4 inhibitors
2.4. Glucokinase activators
Glucokinase (GK) is an enzyme of the hexokinase family that catalyzes the first step in glycolysis [69, 70]. It significantly contributes to the maintenance of the plasma glucose level as a glucose sensor by promoting both liver glucose absorption and pancreatic -cell insulin production [71, 72]. Glucokinase is a phosphorylation enzyme that catalyses the conversion of glucose to glucose-6-phosphate. It is mostly expressed in the liver and pancreatic beta-cells [73]. GK acts as a glucose sensor regulating hepatic glucose metabolism and glucose dependent insulin secretion [74]. GK activators represent a unique and promising strategy to the treatment of type 2 diabetes based on the combined hepatic and pancreatic actions.
Dhanraj et al. synthesized a series of 3, 6-disubstituted pyridine carboxamide [75].
Their anti-diabetic activity was assessed. Compounds 29 and 30 from the synthesised group demonstrated the highest levels of GK activation in the in vitro GK assay (Scheme 7).

Scheme 7. Synthesis of compounds (29) and (30)
Furthermore, the results of the anti-hyperglycaemic activity showed that adding a hydrophobic group, such as trifluoromethyl, to position 3 of the pyridine rings (18 and 19) connected to the carboxamide resulted in improved anti-diabetic activity in the OGTT assay.
Dhanraj et al. created a number of pyridinecarboxamides [76] (Figure 7). With an EC50 value of 1.375 M, compound (31) was identified as the most effective chemical.

Figure 7. Pyridinecarboxamide
It demonstrates that the activity depends on the thiophenyl group being substituted with an ethoxy group at the third position. The substance in question is (DDR63) 3-[(3- ethoxyphenyl)sulphonyl](1,3-thiazol-2-yl) With a predicted activity of 1.375 M, 6-(4H-1,2,4-triazol-3-ylsulfanyl)pyridine-2-carboxamide was discovered to be the most effective compound among the developed compounds.
Yasufumu et al. created a number of 3-pyridylacetamide derivatives with extra hydrogen-bond acceptors that interact with Arg125 in a desirable bidentate manner [77]. According to the findings, compound (32) was identified as the most effective and selective DPP-4 inhibitor that could bind in a special bidentate manner with the guanidino group of Arg125 (Scheme 8).

Scheme 8. Synthesis of compound (32)
2.5. Fuctose 1,6-bisphosphatase inhibitors
Fructose-1, 6-bisphosphatase inhibition is a potentially effective strategy for type 2 diabetes glucose management (T2D) [78]. Inhibiting fructose 1, 6-bisphosphatase (FBPase), a crucial enzyme in gluconeogenesis, prevents the liver from producing endogenous glucose, which has been thought of as a possible pharmacological strategy for managing high blood sugar levels [79].
A number of novel sulfonylureido pyridine series were synthesised by Paul and colleagues [80]. Their ability to inhibit fructose-1, 6-bisphosphatase was assessed. Compound (33) was discovered to be the most effective FBPase inhibitor among the produced compounds, significantly reducing glucose levels and just slightly lowering glycogen levels (Scheme 9).

Scheme 9. Synthesis of compound (33)
2.6. Aldose reductase inhibitors
Aldose reductase (ALR2) converts glucose into sorbitol in the presence of nicotinamide adenine dinucleotide phosphate as the first and rate-determining enzyme in the polyol pathway (NADPH) [81].
The intermediate sorbitol is then converted into fructose by the NAD+ dependent sorbitol dehydrogenase. The majority of the cellular glucose is normally transformed to glucose 6-phosphate, and then processed through the glycolytic pathway by hexokinase.
However, in hypoglycaemia, because of hexokinase saturation, excess glucose is then processed through the polyol pathway, resulting in sorbitol build up and a decrease in NADPH and NAD+.
The imbalance of the NADPH/NADP+ and NAD+/NADH ratios simultaneously affects cellular redox potentials, which triggers the development of hyperglycaemic oxidative stress brought on by both an increase in reactive oxygen species (ROS) build up and impaired antioxidant defence [82]. Moreover, the elevated level of fructose, which is linked to the elevation of the polyol pathway, speeds up the intracellular synthesis of advanced glycation end products and makes it easier to produce more ROS. Antioxidants may lower the ROS concentration and reduce the oxidative stress associated with chronic illnesses, despite the fact that considerably increased levels of ROS cause severe cellular damage when the antioxidant defences are overtaxed [83, 84]. Therefore, inhibiting ALR2 and further reducing the polyol pathway of glucose metabolism are appealing strategies that can stop the advancement of diabetic problems and prevent the aberrant accumulation of sorbitol as well as the damage caused by oxidative stress.
Settimo et al. created [pyrrolo[3,4-c]pyridine-1,3(2H)-dion-2-yl]alkonic acid derivatives using synthetic means [85]. On rat lens enzyme, its in vitro aldose reductase inhibitory activity was evaluated. The compounds 34 and 35 were found to be more effective aldose reductase inhibitors based on the results. The propionic derivatives were less effective than the acetic derivatives (Scheme 10).

Scheme 10. Synthesis of compounds (34) and (35)
Hydroxypyridinone derivatives were created and synthesised by Huan Chen and others [86] (Figure 8). Their ability to inhibit the aldose reductase enzyme was assessed. Specifically, 2-[2-(3,4-dihydroxyphenyl)vinyl] with IC50 values of 0.789 M, -5-hydroxy-4-oxo-4H-pyridin-1-yl-acetic acid (36) was the most effective compound.
Furthermore, 36% demonstrated exceptional selectivity towards ALR2 with a selectivity index of 25.23, which was much higher than that of eparlestat (17.37), the positive control.

Figure 8. Hydroxypyridinone derivative
2.7. G-Protein coupled receptors
Large protein families of receptors known as G protein-coupled receptors (GPCRs) have been investigated as a class of druggable targets for various treatment modalities because they conduct internal signal transduction by detecting chemicals on the cell surface [87][88]. Members of the G protein-coupled receptor (GPCR) family, GPR119, GPR142, and GPR120 have been identified as key targets in the T2DM treatment.
Specifically, pancreatic b-cells and enteroendocrine cells of the digestive system express GPR119. Through Ga coupling, activation of the GPR119 receptor influences adenylate cyclase and raises intracellular cyclic AMP levels [89].
In turn, the pancreatic b-cells are stimulated to secrete insulin. The enteroendocrine cells' expression of the GPR119 receptor promotes the incretins release like GLP-1 and GIP. In addition, endogenous incretins help to regulate blood sugar and may have benefits for b cells [90, 91].
Due to their capacity to lower plasma glucose levels by triggering the actions of this target, pharmacological agents that are strong GPR119 agonists are considered to be viable T2DM regimens.
Novel class of 1H-pyrazolo[3,4-c]pyridine compounds were developed and created by Daisuke et al. [92]. The maximum agonist efficacy among the compounds in this class was found in compound (37), which combined a 4-methylsulfonyl-2-fluorophenyl group (a left-hand substituent) with a 5-ethylpyrimidin-2-yl group (a right-hand substituent) (Scheme 11).

Scheme 11. Synthesis of compound (37)
A series of 6-aminofuro[3,2-c]pyridine-3(2H)-one derivatives of GPR119 agonists were created by Sakairi et al. [93] (Figure 9). The discovery of (38) as a result of further side chain moiety optimization demonstrated robust human GPR119 agonistic action with an EC50 value of 14 M, as well as a delay in stomach emptying and a rise in total plasma GLP-1 levels in mice.

Figure 9. 6-aminofuro[3,2-c]pyridine-3(2H)-one derivative
To boost efficacy and lessen hepatotoxicity, Zhiwen et al. developed a series of new compounds with imidazo[1,2-a]pyridine scaffold as the GPR40 agonist [94]. Compound (39) was discovered to be an efficient agonist, as evidenced by a striking decrease in blood glucose in mice with normal and diabetic blood sugar levels.
Unlike TAK-875, there was no danger of hepatotoxicity with this drug. In addition, the synthesised molecule has good pharmacokinetic (PK) characteristics, with a CL of 27.26 ml/h/kg and a t1/2 of 5.93 h. According to the findings, compound (39) might be a good choice for treating diabetes (Scheme 12).

Scheme 12. Synthesis of compound (39)
As GPR119 agonists, Ying et al. developed and produced 2-(4(methylsulfonyl) phenyl) pyridine derivatives [95]. The two most promising of them, 40 and 41, had EC50 values in vitro cAMP tests of 75 and 25 nM, respectively, and effectively reduced blood glucose excursion in normal mice's oral glucose tolerance test (OGTT). Furthermore, compound (40 and 41) significantly reduced blood glucose levels as compared with the vehicle control group, demonstrating an appealing in vitro and in vivo profile for future research (Scheme 13).

Scheme 13. Synthesis of compound (41)
2.8. Ecto-nucleotide pyrophosohatases/ phosphodiesteases (NPPs)
Nucleotidases found at the surface of cells include ecto-nucleotide pyrophosphatases/phosphodiesterases (NPPs), nucleoside triphosphate diphosphohydrolases (NTPDases), and alkaline phosphatases (APs) [96]. Important NPP family members known as druggable targets for a variety of illnesses, including cancer, type 2 diabetes, and defective calcification are NPP1 and NPP3 [97].
Saif et al. created novel pyrrolo[2,3-b] compounds derivatives of pyridine [98] (Figure 10). The effectiveness of synthetic chemicals as NPP1 inhibitors and NPP3 isozymes, which are overexpressed in diabetes, was examined. The compound (42) with an IC50 value of 0.80 0.04 M was found to be the most effective inhibitor of NPP1.

Figure 10. Pyrrolo[2,3-b] compounds
The most effective and moderately selective inhibitor of NPP3 was discovered to be compound (43; IC50 = 0.55 0.01 M). The key binding site residues of both isozymes, Thr256, His380, Lys255, and Asn277 of NPP1 and His329, Thr205, as well as Leu239 of NPP3, were identified using molecular docking studies of selective and powerful enzyme inhibitors.
2.9. Hepatic glouconeogenesis inhibitors
Fei Ma et al. created analogues of the thieno[2,3-b] pyridine [99]. SAR indicated that adding CF3 to thienopyridine could increase potency and result in the identification of (44) (IC50= 16.8 M) (Scheme 14). The mechanism of (44), which might be effective through the reductive expression of the mRNA transcription level of gluconeogenic genes, including glucose-6-phosphatase (G6Pase) and hepatic phosphoenolpyruvate carboxykinase, was also found (PEPCK).
In addition, (44) could reduce the fasting blood glucose and improve the oral glucose tolerance and pyruvate tolerance in db/db mice.

Scheme 14. Synthesis of compound (44)
2.10. Other anti-diabetic activities
To test for their antidiabetic effect, Tata et al. produced an imidazo [1,2-a]pyridine derivative [100] (Figure11). The positive control, insulin, was used.
It is concluded that the inclusion of a pyridine ring in the second position and N-substituted aryl/alkyl moieties, such as those found in compounds (45) [67.0%], (46) [65.54%], and (47) [64.64%] with substitution., R=cyclohexyl, ethyl, and isopropyl, lower the glucose level in diabetic rats.
In 2007, Rajesh et al. created two series of thieno[2,3-b]pyridines, which were created as BL 11282 analogues, and 2,5-disubstituted 3-imidazol-2-yl-pyrrolo[2,3-b]pyridines [101] (Figure 12). RIN5F cell-based assay was used to assess the in vitro glucose dependent insulinotropic activity of all test substances. One of the chemicals created was 5-chloro-3-(4, 5-dihydro-1H-imidazol-2-yl). Two compounds discovered to be more effective than BL11282 were 2-methyl-1H-pyrrolo[2,3-b]pyridine (48) and 5-Chloro-3-(4,5-dihydro-1H-imidazol-2-yl)-2-methyl-thieno[2,3-b]pyridine (49). This suggests that the pyridine ring of pyrrolo[2,3-b]pyridine and the thiophene ring of thieno[2,3-b]pyridine act as bioisosteres of the benzene and indole ring systems of BL 11282.

Figure 12. Thieno[2,3-b]pyridines
- Conclusion
The main compounds with significant biological applications are pyridine derivatives. These substances cooperate significantly in therapeutic applications. The current review is primarily concerned with the anti-diabetic effects of pyridine and its derivatives to date. A significant issue, diabetes mellitus has led to the development of numerous new compounds that exhibit anti-diabetic activity by either blocking or activating a number of the associated enzymes of disease.
Inhibiting numerous enzymes is a method discussed in this review for achieving anti-diabetic effect, and this review also mentions the enzymes in question. The enzymes discussed in this review are inhibited by a variety of pyridine derivatives, which display anti-diabetic action. The objective of this review study is to highlight the anti-diabetic capabilities of various pyridine derivatives from the research publications that have been published to this point.
Acknowledgment
I want to sincerely thank Sr. Neethu Mathew, for her assistance with the task. I owe a huge debt of gratitude to Dr. Arun Kumar R for his essential advice, recommendations, and support during this work. I have been greatly inspired by his dynamism, vision, and genuineness.
Orcids:
Athulya Chandran
https://orcid.org/0000-0001-5335-1590
Vineesha M.
https://orcid.org/0000-0002-3149-5160
Neethu Mathew Valooran
https://orcid.org/0000-0002-7315-4639
Arun Kumar R.
https://orcid.org/0000-0001-5418-6136
Citation: E. A. Chandran*, Vineesha M., N. Mathew Valooran, A. Kumar R., A Recent Update on Pyridine Derivatives as a Potential Lead for Diabetes Mellitus. J. Chem. Rev., 2023, 5(2), 159-182.


