Document Type : Review Article
Authors
1 Applied Chemistry Department, Adama Science and Technology University, Adama, Ethiopia
2 Chemistry Department, Hawassa University, Hawassa, Ethiopia
Abstract
Non-renewable energy sources produced from natural coal and oil may result in climate change and global warming as greenhouse gases release to the environment. This reason faces the main utilization of energy sources on renewable and alternate green energy to deliver power. Thus, microbial fuel cell device is one of the cost-effective and environmentally friendly energy conversion devices that uses renewable organic wastes as fuel source and converts this stored chemical energy in to useful bioelectricity simultaneously with treating the waste in the presence of biocatalyst. Therefore, it is the most cost effective and simple electrochemical technique that delivers renewable energies. Most of the time electrodes of fuel cells are commercialized and depends on platinum catalyst. Obviously, platinum and its family cost are so expensive. To solve this, researchers are focused on low-cost electrode materials fabrication but still they are in low performance towards wastewater and current generations. Currently, conventional carbon materials are utilized as anode electrodes in laboratory even in pilot scale microbial fuel cell system but are ineffective in their performances due to various reasons. So, to improve performances, anode electrodes were modified with nanocomposites composed from low-cost transition metal oxides and conducting polymeric materials. Therefore, in this review possible ways of transition metal oxide synthesis including green method to form composite with selective conducting polymers, composite modification of anode and its role for wastewater treatment, dye removal as well as bioelectricity generations are briefly discussed.
Graphical Abstract
Keywords
Main Subjects
Introduction
Non-renewable energy sources from coal, oil, and flammable gas, have been utilized to satisfy the world's energy needs for a centuries [1]. However, all of these energy sources are a reason for climate change and global warming [2]. So, the need for renewable and alternate green energy that is regenerated by natural processes as power sources devices from solar, wind, biomass, hydropower, geothermal, and fuel cells (FCs) are the main utilization energy sources to deliver power [3]. Among, FCs is an electrochemical cell that converts the stored chemical energy of fuel into electricity. It contains both electrolyte and two electrodes (anode and cathode) in which a pair of redox reactions is the main process during the conversion process. Therefore, it is the most cost effective and simple electrochemical technique that delivers renewable energies. Hence, renewable energies produced through these entire devices have the potential to limit our dependence on hydrocarbon fuels, but its startup time, low power output, short service life and high cost limits its commercialization at large.
As an advantage FCs has a supply of the fuel to produce clean energy and can be used in various small devices [3]. Most of the time electrodes of FCs are commercialized and depends on platinum catalyst. Obviously, platinum and its family cost are so expensive. To solve this problem, researchers are focused on low cost electrode materials fabrication but it isn’t successful and still they are in low performance [4]. So, in this review, electrodes composed from low cost nanomaterials from metal oxides, conducting polymers, and their composites as a catalyst, using fuel from organic waste products rather than using hydrogen as a fuel and the simultaneous liquid waste treatment are discussed in brief. Nanomaterial’s synthesis, characterizations, and their uses renewable energy production from liquid wastewater and its simultaneous treatment using microbial fuel cell is clearly summarized.
In bioelectrochemical system such as microbial fuel cell (MFC) promising electrochemical cell devices in the FCs system that produces a bioelectricity renewable sources of energy from stored chemicals found on the organic matter pollutants in the presence of electroactive microorganisms (EAM) [5]. In principle, organic substrates are oxidized on anode electrode while oxidizing agents are reduced at the cathode [5,6]. MFC is in this way characterized as the device to create renwable bioelectricity from stored waste effluents found in the environment through the action of dynamic microorganisms which act as a biocatalysts [7]. The way to produce bioelectricity from such organic effluents and green treatments of liquid wastes itself have been received much attentions in bioelectrochemical system [8]. This devices is a key strategy to reduce energy crises and to keep the environmental safety [6]. Due to this reason, emphasis has been given to MFC system beyond its technological novelty [9]. To prove this research advancements have been reviewed on bioelectricity with simultaneous waste treatments from various waste discharges [5, 7]. However, a resistance found on the anode electrodes limits waste treatment and renewable energy production efficiencies [10]. Such huge problem is a cause for cost ineffectiveness due to slow electron transfer mechanism between EAM and anode surface [11]. To solve this challenges, creating an effective contact between active microbe and solid conductor through physical, chemical, electrochemical techniques play a vital role [12,13]. To summarize all possible factors and its possible reduced mechanisms by using nanomaterials composed from selective metal oxides and conducting polymers are explained below.
Anode electrode role in MFC performance
In order to minimize all limitations found on anode electrode, fabricating it with superior material having with better specific surface area with good roughness, good in electrical conductivity, good in chemical stability, and best in biocompatibility could be given as the primary concern in MFC device system [14,15]. Still, it is a problem to introduce highly effective microbial supportive bioanode materials [16]. The supportive materials are clearly helps for the growth of EAM biocatalyst. When successive feedings of substrate is loaded in to anode bioreactor it causes the maximum power densities increased due to the adaption and growth of bacteria in the anode chamber [15]. If this criteria’s are fulfilled, it is expected that oxidation rate of organic compounds found on anode chamber MFC is faster and the cell performances is improved [17]. To support this, researchers have been primarily focused on low-cost conventional carbon electrode materials [18] due to their commercial availability [19]. But most conventional carbon electrode have poor chemical stability, small surface area, large pore size, poor mechanical stability, and low degree of biocompatibility [20]. As a result, electrodes from these conventional carbon materials have a limited catalytic activity during the course of the reactions at MFC. They also demonstrate low oxygen reduction activity due to their high over-potentials at low temperature and neutral pH [13]. For example, pencil graphite anodes (PGE) is quite possibly the most industrially accessible carbon material because of its, reproducibility, sustainability, superfluity, and its accommodation during time of activity [21–23]. In any case, PGE and other most traditional carbon anodes actually have limitations, like low electrical conductivity, poor biocompatibility, and restricted surface response destinations, which would slows microbial growth rate and this causes the sluggish electron transfer rate and decreases MFC performances [24]. So, conventional carbon anodes needs surface modification that changes the electrochemical behavior of MFC beside improving its conductivity to reduce the ohmic resistance which is the dominant factor that affects MFC performances [25]. Therefore, modifying this electrodes with functional hybrid composites derived from inorganic-organic integrations is expected to increase its bioelectrochemical catalytic activity [26–29]. So, modification of conventional carbon based anode with smart-functional nanomaterials composed from metal oxides and conjugated conducting polymers [30] was effective to achieve performances in MFC [31,32].
Among many, some of the conventional carbon based electrode materials from HB-pencil stroked graphite [33], black and activated carbon [34], reticulated vitreous carbon and graphite felt [35], carbon fiber [36], graphene foil [37], and toray carbon paper [38] have been reported as a common and selective anode electrodes in MFCs. However, their poor electrical conductivity, inefficient bacterial adhesion, and low bioelectrocatalytic redox activity limits the electron transfer rate between active microbe and anode [39]. To improve these drawbacks, modification of those carbon materials with transition metal oxides (TMOs), and conducting polymers based nanomaterials, such as MnO2 and Fe3O4 on carbon black [11], MnO2 on carbon felt [19], iron-plated carbon-felt [40], FeO nanoparticles coated carbon [41], polypyrrole (PPy) [42], sulfuric acid doped polyaniline (PANI) modified carbon cloth [43], PANI-modified glassy carbon [44], and their composites, such as MnO2/PPy/MnO2 [45], multiwall carbon nanotube/MnO2/PPy) [46] on carbon cloth, poly(3,4-ethylenedioxythiophene)(PEDOT)/NiFe2O4 on biochar [47], PANI/TiO2 [48], polyaniline/molybdenum trioxide/activated carbon cloth (PANI/MoO3/ACC) [49] have been done for better MFC performance.
So, improving the surface of anode with various methods in the presence of active functional nanomaterials as a catalyst enhances electroactive bacteria community followed by increasing rate of electron transfer. Such improvement enhances MFC efficiency [13,25]. During electrode modifications both electrical conductivity and surface areas might also improve and this minimizes the charge transfer resistance found in the cell [25]. Therefore, it is interesting that an electrode could increases the energy conversion efficiency of the cell when an anode of the cell devices modified from a mixture of TMOs and redox polymers. Nanomaterials with good performance have a versatile and superior properties corresponding to the bulk counterparts [50]. To reduce all the challenge explained above, using nanomaterials as a catalyst could be selected as a multifunctional material for renewable energy production and waste treatment due to their best electro-catalytic activity, high thermo-chemical stability, elasticity, and many others. Such properties in an energy conversion device allows for liquid waste treatment and power conversion efficiency enhancement [32] by reducing charge transfer resistance found at electrode/electrolyte electrochemical reactions.
By providing the above criteria, Phonsa et al [51] have reported a liquid waste treatment removal efficiency of 100% when a bare anode was modified by MnO2/PPy composites through electrodeposition method. After a test, the formed power conversion efficiency was rich to 440 mWm−2. From this report, the produced power is still too low and not sufficient to power an electronic device that limits their use in commercialization applications. In the same manner, other reports are also briefly summarized from the following sections. Especially, the role of TMOs based composites with its green preparation strategy and its electroctalytic activity role as a catalyst modifier to anode in MFC efficiency improvement could also discussed.
Transition metal oxide catalyst activity in MFC
Recently, TMOs based nanomaterials have been emerged as a promising nanocatalyst to improve anode in MFC performance [26]. The best in structural defect, stability, and availability [52] could attract its unique applications in the electrochemical catalysis system [53]. Therefore, its excellent structural dynamicity, environmental friendship, and nontoxicity properties makes TMOs is unique to modify electrodes [54]. So that TMOs based nanomaterial is suggested as a good candidate for anode modifier catalyst in MFCs [6]. In particular, due to having unpaired d-orbital electrons on their structure enabling their electrochemical catalysis activity in MFC [55, 56]. To increase its chemical, mechanical, and electrical conductivity, it needs further modification with other novel and active nanomaterials like conducting polymers [30] would be able to decrease charge transfer resistance [57]. Compositing of metal oxides with conducting polymers appeared to be a promising material for an electrochemical catalytic activity in MFC. After preparing them by facile synthesis, good electro-conductivity, environmental stability, fast electro kinetics exhibition, excellent catalytic activity, lower toxicity, and redox stability of conducting polymers [45, 46]. That is why; combinations of inorganic nanomaterials with redox polymers have been attracting a considerable attention in many fields of applications. So, in this review modification anode electrode with some basic green synthesized TMOs based composite conductive materials leads to facilitate electron transfer rate by decreasing the associated energy losses formed at the interface between biocatalyst and electrode in the MFC device system. The detailed summary of plant extracts and its role for some TMOs nanoparticles and their composites are also highlighted.
Plant extracts role for TMOs based nanoparticle synthesis
An extensive biological sources from plant extracts and microorganisms help to synthesis nanoparticles from their metal precursors [58]. Preparing them by using an active microorganism as a reducing agent is not cost effective and is not an easy task for commercialization. To solve these problems, emphasis are dominated on plant based synthesis due to ease of improvement, i.e., it eliminates the elaborate process of maintaining cell culture, safe to handle with a full range of metabolites and other active groups, ease of its simplicity to avoid complex mediated synthesis (multi-step reactions) [59]. Based on such criteria, plant assisted nanoparticle preparations have captured a considerable attractive interest in the areas of modern nanotechnology [60]. Preparation of efficient TMOs nanocatalyst with chemical wet methods are highly affect the environment safety due to the corrosive nature of chemicals which were used as a reducing agent during synthesis. Therefore, finding safe, simple, green, and alternative to the wet chemical method are highly encouraged to keep the safety of lives found on the ecosystem. Therefore, preferring a plant lead nanoparticle synthesis method is chosen as a green and time saving approach [61].
So that the biosynthesized approach is more preferable and alternative way due to its safeness, environmentally friendly nature, non-hazardous effects, and ease of synthesis [62–64] but all these requirements are not found in other wet-methods create some [65]. So, the need to minimize such toxic reagents and to develop a well-dispersed nanoparticles using plants extracts are a well-established strategy in the current research worldwide [66, 67]. Phytochemicals that extracted from leaves, flowers, roots, stems, and seeds have a reducing and stabilizing agent activities as reported from many research works [63, 68, 69]. Their primary reductanct activity nature they enhance nanoparticle reducing capability from their metal precursors [70] of plant extracts, various metal oxides nanoparticles have been well prepared [71]. In addition to their stabilizing ability, extracts have also a role to prevent nanoparticles from surface aggregations which is commonly known as capping agent [72]. The presence of dense aggregation reduces the reactive sites of the active nanoparticles which then reduce the active surface area [59].
It is reported that, plants contain various kinds’ phytochemicals from terpenoids, organic acids, and many others. It has been elucidated that those organic compounds contains an active functional groups, such as hydroxyl, aldehyde, amine, and carboxyl units, which are a primary source of electrons [73] and could increase the reduction and stabilization of nanomaterials [74]. Furthermore, plant derivatives are believed to be superior in accelerating metal ion reduction rate compared to microorganisms, and better for producing metal oxide nanostructures with distinct morphologies and improved stability [72]. However, preparation of TMOs nanostructures by using plant-derived phytochemicals typically requires longer time during extraction, isolate, and purifications of the phytochemicals and the follow charts that showed their extraction are shwn in Figure 1. Finally, the TMOs nanostructure intrinsic properties, green-synthesized metal oxide materials have a role for environmental remediation and energy conversion/storage applications. It is important that natural agents can used to replace hydroxide ion source coming from different chemicals, such as NaOH, NH4OH, CO (NH2)2, and hexamethyl tetraamine. In this mechanism, hydroxide ion sources are usually required as catalysts to accelerate the nucleation and formation of metal hydroxides, which in turn leads to easier formation of metal oxides. Previous studies reported that biomolecules are cost effective and safe catalyst for metal oxide formation.
.%5B2%5D.jpg)
Figure 1. Schematic diagram showing the general steps for obtaining phytochemicals from plants [72]
For example, Gunalan et al [75] have reported CuO nanoparticles from CuSO4 precursor using Aloe barbadensis Miller extract to replace hydroxide ion coming from NaOH chemical. The synthesized nanoparticle was spherical in shape with sizes between 15-30 nm as shown in Figure 2a. Aloe vera extract has also been used to synthesis of ZnO nanoparticles. It is interesting that 90% of nanoparticles yield was achieved using Aloe vera extract while only 50% yield nanoparticle was achieved with NaOH. Even the ZnO synthesized nanoparticle reaction time by using Aloe vera was faster than using NaOH. The average size of ZnO nanoparticle is 35 nm and its surface topography is clearly revealed in Figure 2b. Therefore, it is understood that from the synthesize of both CuO and ZnO nanoparticles were promoted by either vitamins, enzymes, minerals, sugar, lignin, saponin, salicylic acid, or amino acid biomolecules found on Aloe vera plant [72]. It is important that the free amino acids and carboxylic groups present in phenolic biomolecules were thought to be responsible as a capping agent (i.e., protecting nanoparticles aggregation).
.jpg) Figure 2. TEM images of (a) CuO and (b) ZnO [72]
Figure 2. TEM images of (a) CuO and (b) ZnO [72]
In addition to CuO and ZnO nanoparticles, the biosynthesis of MnO2 nanoparticles from different types of plant part extract have been reported and its optical properties as well as its average crystallite sizes are compared and given on Table 1. Generally, it is concluded that the roles of plant extract are primarily used as a reducing and stabilizing (capping) agents.
Table 1. As synthesized UV-Vis maximum wavelength and average crystallite size comparison of calcined MnO2 nanoparticles [64]
.jpg)
These novel enhancing nano-sized preparation from different metal such as Mn, Fe, Co, Ni, and etc. oxides are some of the most common and cost effective super-catalytic materials [78]. Their phase, size, and surface topographies on these nanomaterials have also a great influence on their applications. Therefore, several novel and effective routes have been devoted to prepare those nanomaterials with various shapes and excellent properties. Composites from this metal oxides have been attracting due to their mesoporous dominancy, better stability, flexibility, and widespread functional properties which would help to play their key role in numerous industrial, biological and analytical fields [79]. As a result, their combinations have had better porosity in their frameworks. This is happened due to their tunable properties upcoming from their multiple metal species together. These are revealed by the significant contribution and synergetic role of many oxide composites in numerous applications where individual single-component oxide fails to yield the necessary outcome. So, combination of these materials expected to develop super-functional with new features in the resulting material which is different from the individual one [79]. Based on this prospective, some binary metal oxide-base nanomaterials and their composites as well as their synthesis mechanisms are reviewed effectively.
In the oxide composites, oxide-oxide and oxide-silica composites are the two major varieties. This is due to having high chemical stability, the flexible tetrahedral building blocks of silica, huge surface area, and tunable porosity facilitate the easy synthesis of silica and silica-based oxide composites making them extremely significant in many promising research areas. The high susceptibility towards chemical hydrolysis, oxidation, and difficulty to retain porosity upon thermal treatment makes these syntheses of oxide-oxide porous composites quite challenging. To solve such problems, the addition of ions or compounds into a basic matrix at a very small concentration usually called doping should be governed. The ratio of the additive to the basic material known as the host must be very low and the powder XRD analysis may or may not show the peaks of the doped species but there will be a shifting of the XRD pattern for the host material [79]. On the other hand, due to its cost effectiveness and environmentally friendly nature, green synthesis on bimetallic oxide-base materials other than silica-based oxide composites have been successfully reported by using green preparation method with plant crude extract as a safe reducing agent. Among this, CoO-NiO [80], Pd/MnO2 [81], Cu/MgO [61], and Cu/MnO2 [82] are some of them. To clarify the reducing ability of plants, an electrochemical redox mechanism between the plant extracts and metal-based precursors have been explained as follows. Such mechanism is a good model for biomolecule reducing ability extracted from different parts of plants [83]. For example, Maqbool et al [84] used O. europaea extract as both reducing and capping agents for the nucleation and growth process of CeO2 nanoparticles with a uniform spherical shape and mean size of 24 nm [84]. Later Maqbool has reported the excellent biocompatibility of green-synthesized CeO2 nanostructures [85]. This is due to that the founding of oleurepein and oleuroside biomolecules as the main constituents in O. europaea extract. During the biosynthesis process, oleuropein was decomposed into elenolic acid and hydroxytyrosol. Both of them can act as an organic capping agent and the reaction mechanism is demonstrated in Figure 3. According to the report, the presence of other biomolecule constituent which is found in the extract as an organic impurity did not influence the crystal growth of CeO2 nanoparticles. So, the final average crystallite sizes are 18 nm and 26 for CeO2 and Ag nanoparticles, respectively [85].
.jpg)
Figure 3. Capping agent role of O. europaea extract in the crystal growth of CeO2 and Ag nanoparticles [85]
TMOs based nanocomposites have the potential to increasing stability and super-catalytic activity of electrode materials in electrochemical redox reactions [86]. Inexpensive TMOs based nanocomposites are a promising and alternative electrocatalyst to replace carbon supported platinum catalyst electrode due to its expensiveness. So, this cost-effective composites have a great benefit in oxygen reduction reaction with in energy conversion devices. Khan et al [87] have reported a cathode catalysts modifier from MnO2 nanoparticles in air-cathode cubic MFCs. In this device anodic chamber was inoculated by anaerobic sludge and palm oil mill effluent was used as a substrate. A maximum power density of 165 mW m-3 was produced by MnO2 modified Pt cathode while only 95 mW m-3 power density was produced with MnO2 nanoparticle catalyst [87]. From this study, the role of anaerobic sludge was used as a complex biocatalyst that are increasingly examined as an alternative approach for wastewater treatment and soil remediation after decomposing organic pollutants beyond bioelectricity generation [88]. Continuously, the performance role of composites from TMOs in terms of power and current density efficiencies in MFCs device system are reviewed in Table 2. From the review possible composite synthesis methods are given.
Table 2. Summary of MFC polarization parameters [28]
.%5B2%5D.jpg)
Conducting polymer role for anode in MFC performance
Among, PANI is one of the most attractive conducting polymers among other redox conducting polymers in the electrochemical studies due to its low-cost, good biocompatibility, and environmental stability properties [95]. But its inefficient conductivity of PANI in neutral aqueous solution often limits power generation and its simultaneous wastewater treatment in any types MFC [96]. Since, MFC is operated at optimum conditions either neutral or weak basic solution but PANI at this experimental condition is unstable due to structural deformation and shrinks [97]. As an advantage, PANI carries a positive charge on its backbone, therefore, when negatively charged EAM outer cell membrane interacts with such charge, they lead to form a conductive biofilm on electrode surface due to strong adhesion of more EAM coliforms resulting from strong synergistic effect between polymer and bare electrode materials [49]. Such opportunity increases the anode electrode performances by increasing its specific surface area then promote electron transfer rate across the formed interface, and finally improves the whole electrochemical activities after effective surface modifications [97]. Due to this reason, various research have been working on modification of carbon electrode by PANI combination and this be able to enhance electron transfer rate between EAM and electrode surface [98–100]. Next to PANI, poly (3, 4-ethyldioxythiophene) (PEDOT) is also a noteworthy conducting polymer that used to modify anode electrode surface to due to the presence of delocalized π-electrons at the polymeric chain backbone that enable to induces unique optical and electrical properties, such as electrical conductivity, good biocompatibility, environmental stability and simple synthesis procedures. Based on this intention, Hernández et al [14] have performed PEDOT based graphene/nickel nanocomposites modified anode by using electrochemical polymerization technique. Their ease of preparation and application mechanism is illustrate in Figure 4. Its performance ability was done in the presence of Escherichia coli biocatalyst feed with glucose as the substrate and its absolute power and maximum power density were 3.9±0.3 mW and 0.32 mW cm-2, respectively.

Figure 4. Schematic representation of a microbial fuel cell and direct electron transfer between electroactive biofilms and modified electrode (where, Np-Ni = Nickel nanoparticles, ErGO = electrochemical reduced graphite oxide, E. coli = Escherichia coli) [14]
In particular, modification of MnO2 by PANI to form hybrid composite and coated on PGE would make the best opportunity to addresses the low performances in MFC. This strategy is succeeds by low-cost MnO2 but MnO2 is highly unstable and is poor in electrical conductivity at protonic media [6]. At this media, it changes its phase and decreases its performance in MFC. In the +4 oxidation states of MnO2, α-type chain-like tunnel structure (α-MnO2, 2 x 2 tunnel [101]) exhibits fast electro kinetics and excellent catalytic activity in the neutral electrolytic solutions due to having a large surface area and large number of reactive surface sites and has the advantages of being environmentally friendly and abundancy beyond its cost effectiveness [45]. In addition to modification of conventional carbon anode with hybrid composites, configuration of MFC device system has also an impact on its performance. Any of the device setup has their own advantage and disadvantage [102]. Since double chambered MFC has an advantage over single chambered MFC configuration. For example, purging of oxygen in single chambered configuration might cause direct oxygen interference due to cathode insertion in a common electrolytic medium. The diffused oxygen towards anode bioreactor might be consumed by biocatalysts and is a cause for low substrate oxidation. Although there have been some challenges in using air cathode, the sluggish kinetics during the oxygen reduction reaction in air-cathode causes the low extracellular electron transfer efficiency through outer-membrane bound cytochromes, redox mediators [7] depicted in Figure 5, resulting in poor MFC performance [103]. Anode modifications using nanomaterials in single chambered MFC might give rise to electron diffusion resistance between the interface [104]. As a result, choosing a flexible dual-chambered MFC set-up have been given a broad emphasis to solve this problem [105].
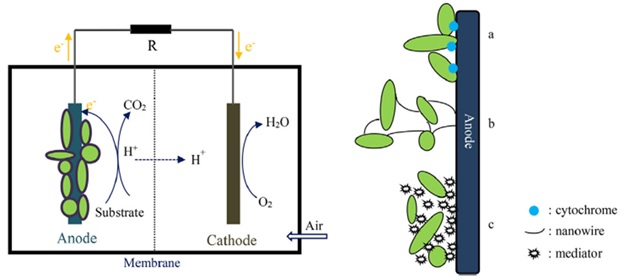
Figure 5. MFC illustrations and electron transfer mechanism (a) outer-membrane bound cytochromes, (b) redox mediators (c) redox mediators [7]
Based on those detailed observations, a novel with biocompatible hybrid composite materials from metal oxide/conducting polymer matrix have prepared by chemical polymerization means to increase anode catalytic activity in any substrate fed based MFC device system [20]. For example, single chambered MFC performance test was done in the presence of EAM biocatalyst for bioelectricity conversion and waste recycle. In detail, the presence of active microorganisms with different metabolism process like fermenters, methanogens, or electrogens, is possible to decompose organic matter that is present in discharged wastewater into simpler compounds that are used by electrogenic species for electrical current production. Through this decomposition the generated current is obvious. It is interesting that, wastewater treatment with MFC is not only allowed for organic matter removal but also for reducing content of nitrogen, phosphorus, and various, often toxic, metals like Cr, Cu, Fe, and Co in wastewater. It is common that for some wastewaters, COD or metal removal efficiencies exceeded 90% what enables direct reuse of the MFC-treated water [106]. For such application, PANI exhibits good environmental stability and high conductivity, specifically as a simple and reversible acid/base doping/de-doping chemical property [107]. The change in colour found in PANI is due to the existence of the polymer with different states of oxidation and these existences are very useful to increase the sensing device of the cell. Although color is also useful, the best method for making a PANI sensor is to take advantage of the dramatic changes in electrical conductivity between the different oxidation states or doping levels. PANI is more noble than copper and slightly less noble than silver which is the basis for its broad use in printed circuit board manufacturing (as a final finish) and in corrosion protection [108]. So, to improve the performance of PANI, it has been recognized to make composites for electrode materials designing in the electrochemical device system. It has a usual benefit to increase electrical conductivity and biocompatibility by formic high surface area and surface roughness towards bacterial attachment in MFC anode [93]. So, to overcome all limitations found on metallic oxides, researchers usually combine them with conducting conventional carbon materials and conducting polymers to improve conductivity, compatibility, and dispersibility [109–111].
The effective electrical activity of conducting polymer coated on anode enhances conductive biofilm which facilitates bioelectrochemical reactions in MFC. Thus, the formed bioanode materials play a significant role in MFC performance. This is effective when a desirable anode should have enhanced conductivity, higher specific surface area with good surface roughness, good biocompatibility, and reasonable stability [112]. So, to increase anode efficiency further, conducting polymers modifications with different metal oxide-based composites could offer a great opportunity to upgrade the required electrode in MFC performances. For example, PANI/carbon felt [113], NiO@PANI/carbon felt [94], PANI/chitosan/graphite rods [114], MnO2/PPy/carbon felt [89], MnO2/PPy/Stainless steel [115], PANi and PPy/Stainless steel [116], ferrite (MnFe2O4)/PANI/carbon cloth [117], Nickel oxide/carbon nanotube/polyaniline/carbon cloth [93], etc. Therefore, conjugated polymer-based metal oxide nanoparticles coated on conventional carbon electrode is a good and fantastic option to improve power production and toxic compound destruction. To prove this when polypyrrole (PPy) was coated with MnO2, Fe2O3, and MnO2-Fe2O3 nanocomposites were done by modifying anode surface and improve performances in benthic MFC as demonstrated in Figure 6. Among these, Fe2O3/PPy exhibited higher power density (170 mW/m2) than that of other MnO2-Fe2O3/PPy (117.29 mW/m2), MnO2/PPy (90.54 mW/m2) and unmodified electrodes (69.19 mW/m2). According to the study, the properties and performances of nanostructured modified anodes were studied by measuring their electrochemical behavior and power generation capacity. The Fe2O3/PPy coated anode exhibited 409 times higher kinetic activity than control in this benthic MFC. This is due to their best in surface area, and the presence of rich electron donating functional groups and heteroatoms [118].

Figure 6. Polarization curve obtained from the reactors: (a) V-j curves, (b) P-j curves [118]
Finally, the nanocomposite combined from different metal oxides with conducting polymers and their efficient bioelectrocatalytic activity towards bioelectricity generations is summarized in Table 3.
Table 3. Bioelectricity production and COD removal efficiency produced by different composites modified electrode in doubled MFC [5,6,27]


GNRs = Graphene nanoribbons, GCE = Glassy carbon electrode, TA = tartaric acid, GR = graphite rode, BC = Bacterial cellulose, CNTs = Carbon nanotubes, APTES = Υ-aminopropyltriethoxysilane, ITO = Indium-tin oxide, PEM = Proton exchange membrane, Antimony-tin (Sb-Sn), PGE = Pencil graphite electrode
Role of MFC for bioremediations
In MFC design, considerable efforts have been done to enhance its performance towards wastewater treatment [91]. It was stated that low power density output, poor stability, and cost-ineffectiveness are the main drawbacks that affects the practical advantages of MFC [92]. The low electron transfer efficiency of electrode reduces the MFC energy conversion performance and its bioremediations [32,92]. Bare electrodes contained from carbons are poor in remediation applications due to poor long-term stability results for slow electron transfer rate between electrode and bacterial cell [125]. This is clearly visualizing the limiting factor for the practical applications in MFC. To solve these limitations, integrations of nanostructured materials from various types for remediation activity have been reviewed so far [20]. Money nanoparticles from TMOs have been utilized as effective catalyst in energy conversion and storage applications due to its low-cost, environmental friendly nature, abundance, and high the catalytic activity [57]. The founding of various types of crystallographic forms in TMOs nanoparticles makes them a significant role for fast electrokinetic activity in wide spread of electrochemical uses [5].
Among, α-MnO2 NPs have drown a considerable attention due to its good pore structure surface and large reactive surface area [64]. The better chain like tunnel structure found in α-MnO2 NPs makes a better bioanode catalyst modifier in MFC [5]. Its poor electrical conductivity, poor dispersability, and low stability in highly protonic media reduces the long term electrochemical treatment efficiency [6,57]. As an effective bioremediation, compositing of α-MnO2 NPs with NiO and redox PANI exhibited a good electrocatalytic activity to wards extracellular electron transfer rate between conventional carbon material and electroactive bacteria. However, the low electrical conductivity in NiO [93] and lowering life cyclic during charge/discharge processing time in PANI [126], could restrict long term waste treatment efficiency in MFC performance. Since TMOs in general have high theoretical capacitance and be a good option anodic material when they makes hybrid with conducting polymers due to efficiency and stability modifications [94]. As result, when the conventional carbon electrode was modified with nanocomposites, it should meet large surface area, durability, stability, and porosity requirements to facilitate fast extracellular electron transfer efficiency between active microbe and solid electrode by forming an immobilization matrix [126]. That is the immobilization of electro active bacteria with the help of composite catalyst enhance the close contact between bacterial cell and electrode surface. Therefore, the synthesized nanocomposites enhances electron transfer movement within the MFC device.
Under the umbrella of conducting polymers, due to its simple ease of synthesis and electrochemical catalytic activity, PANI could help to reduce extra cellular electron transfer resistance in MFC [6], but this activity is reduced in the neutral and near neutral media due to inefficient electrical conductivity [96]. As a result, it needs its surface modification. In addition to conducting PANI, MnO2 has also an efficient electrocatalytic activities due to its diverse physicochemical properties. Thus MnO2 based hybrid composite modified conventional carbon anode electrode with different conducting polymers, such as polypyrrole/MnO2 [92], MnO2/polypyrrole [89], MnO2/PANI/MnO2 [100], MnO2/Polypyrrole/MnO2 [45], multiwalled carbon nanotubes/MnO2/polypyrrole [46], were effectively studied to improve MFC performance. It is extended that, to solve instability, high cost, and lower power efficiency followed by low treatment efficiency would be able to give the intention to prepare ternary based composites in MFC applications [126].
Provided that substrate type and concentration are major factors that affect bioremediation in MFC. Until now, substrates from several organic compounds such as domestic wastewater, industrial wastewater, winery wastewater, and waste activated sludge have been tested effectively. Anaerobic activated sludge which is taken from the aeration tank in any wastewater treatment plant biomass and recycled through the reactors until current is generated in the bioelectrochemical system. It is used as the inoculum in MFC to enrich the biofilm at the anode surface to contribute a conductive biofilm. In a MFC inoculated with anaerobic activated sludge and fed continuously with artificial wastewater containing glucose and money, othor organic compound would find in the anodic bioreactor. Anaerobic sludge’s from a biological wastewater treatment plant constitutes a good source of exoelectrogenic bacteria and can grow up to 35oC [127].
MFC is one of the most prominent device in the bioelectrochemical system applications, especially for bioenergy, bioremediations, dye removal from liquid waste, batteries, and even for biosensors [92]. In this review, only bioenergy (current generation), bioremediations, dye removal from liquid wastes were discussed on the following sections.
Wastewater treatment and bioelectricity production
It was explained that electrochemical optimization of anode using functional TMOs and conducting polymer materials can occur through enlargement at the specific electrode surface or insertion of nanocatalytic mediators in either of electrodes or in the feeding solution [42,128]. Enhancement of electrochemical performance with TMOs based nanomaterials towards oxygen reduction reaction and their output potential and power density was much better than most conventional carbon materials in neutral media but it has high activation overpotential under similar conditions [129]. Martin et al [130] reported Mn2O3 nanopowders modified carbon black electro catalytic activity with a COD of 86.5% under air and oxygen atmospheres in air-cathode MFC under mesophilic sludge effluent. The equivalent open circuit potential produced by this catalyst was reached to 844 mV vs Ag/AgCl [130]. To facilitate EAB biofilm formation and its cellular electron flow to the anode, Sarma et al [131] have synthesized Fe3O4 encapsulated with aniline and pyrrole composite polymers. As a result, the produced open circuit potential was reached to 0.949 ± 0.07 V with COD removal of 65%. In this fuel cell acetate was used as a fuel on the prepared artificial wastewater that produced a current density and power density of 14.04 ± 5.5 A m−3 and 4.9 ± 0.5 W m−3, respectively. The conducting polymers which were coated on Fe3O4 as a composite anode modifier was better in biofilm formation. This enable to treat wastewater effectively and enable to produce high power output in the MFC [131].
Stainless steel (SS) due to low cost and high mechanical strength is regarded as an eligible and preferred anode material to treat wastewater in MFCs. However, its poor biocompatibility and low corrosion resistance still limited its treatment efficiency. Pu et al [4] have improved its performance by modifying it with PPy. Therefore, after modification its power density was reached to 1190.94 mW m−2, which was about 29 times higher than bare SS anode. After modification of PPy/SS by MnO2 through electrodeposition its wastewater treatment efficiency was 100 % of COD while its power output was too low with a value of 440 mW m−2 [51]. On the other hand, Chen et al [89] reported on MnO2 based PPy composite anode by in situ chemical polymerization method. According to their report the wettability test from the contact angle measurement was reduced to 46±0.5°. Finally, its maximum power output density was reached to 562.7±10 mW m-2. Hence, the reduction of contact angle would promote bacteria attachment followed by increases catalytic activity and this reduces the electron transfer resistance, which is the basic requirement for modified anode performance. This leads microbe adhesion improvement which can be attributed to its hydrophilic property of the modified anode, which is consistent with its smaller contact angle and better wettability [89].
Haoran Yuan et al [45] have explained that when a carbon cloth was decorated with inexpensive hierarchical multi-walled high-performance MnO2/PPy/MnO2 nanocomposites, its maximum power density was reached to 32.7 ± 3 W cm-3. This value was 1.3 times higher than that of unmodified carbon cloth with increase in durability. Impedance measurements evidenced that the modified anodes lead to a low charge transfer resistance and provide unique active center to host the bacteria for more efficient electrocatalysis towards substrate oxidation (waste decomposition). To test the application, the anode bioreactor was composed with 1000 mg L-1 sodium acetate solution in 50 mM phosphate buffer containing 10 mL.L-1 mineral solution with 10 mL.L-1 vitamin solution [45]. It is important that MFC were constructed and effective output was obtained with binder-free NiO/MnO2 composite modified carbon felt anode by using dye containing wastewater. Here, the composite was prepared with a facile two-step hydrothermal method [91]. By using the same conventional carbon felt, polymerization of PANI embedded NiO was grown on bare carbon felt electrode to form NiO/PANI/CF. with this composites, a maximum of 1078.8 mW m-2 and 64.24% power density and COD removal efficiencies after 48 hour scan time were generated, respectively [94]. A nanocomposite was also prepared from nickel-nickel oxide (Ni-NiO) nanoparticles (NPs) on reduced graphene oxide (rGO) using pyrrole (PPy) as a support matrix. According to the report, a higher current density and power density of 2134.56 mAm-2 and ~ 678.79±34 mWm-2 in comparison to the commercial Pt/C catalyst (1788.2 mAm-2 and ~ 481.02±24 mWm-2) was produced by this cost-effective Ni-NiO/PPy-rGO nanocatalyst. Its respective removal efficiency in terms of COD was reached to 81.52% over 30 days of MFC operation. Being an electrically efficient catalyst, these combinations can be also optimized for other known conventional catalysts in MFCs [6,132].
An electron mediator material modified with conductive polymer-based nanocomposites could also consider as a suitable bifunctional anode catalyst for high-performance MFCs for sustainable energy production and wastewater treatment application. Nourbakhsh et al [93] have used NiO as effective electron mediators in CNT and PANI by forming composites (NCP). When these composites modified carbon cloth, it facilitates an electron transfer rate between bacterial cells (Shewanella Sp.) and carbon cloth electrode combine composites due to effective synergetic correlation formation as demonstrated in Figure 7. Other than TMOs, wastewater treatments by using co-polymerization from conducting polymers have also the role to keep microbes safety. Composites from conducting polymers from PPy and PANI polymeric electrolytes as a co-polymer was used as a good anode modifier in MFCs. For this proof, when SS wool is coated with PANI-co-PPy, their power density was improved by 70% of bare SS wool alone under similar conditions. In this applications, acetate and landfill leachate were used as a fuel and bacteria sources, respectively [116].

Figure 7. Anode surface modification by NCP and its electron transfer mechanism and interactions among of modified NiO/CNT/PANI/carbon cloth anode and Shewanella sp. Bacteria [93]
Dye removal and current generation
As an efficient method, the development of bioelectrochemical device systems like MFCs have been emerging as a simultaneous alternative method to degrade dyes and generate current through electroactive bacteria as a biocatalyst [133]. Nowadays, this technique is receiving much more attention than other previous conventional treatment methods due to its innovative and eco-friendly nature. Still the dye removal efficiency was quite low due to the use of conventional carbon-based electrodes. However, it is obvious that modification this electrode with functional nanomaterials offered higher dye removal efficiencies due to surface area modification which enable to provide more active bacteria attachment. Such modification increases the pollutant removal efficiency due to an increase in surface reactive site improvement. So, the effect of the electrode was very substantial for both dye removal and current generation. Producing power from huge dye containing organic waste effluents and keeping the environment safe is the basic strategy given by all researchers. So, producing a renewable energy from those waste effluents containing azo dyes have been proposed as an alternative approach to improve the economic efficiency [114]. Azo dyes discharged from textile, paper, and cosmetic manufacturing are highly toxic to the environment. The continuous discharge of this dye compound containing wastewater affect human health and ecosystem due to their toxicity and bioaccumulation [134]. Physical, chemical, and biological methods have been carried out to treat a wastewater containing from azo dye containing organic compounds [134,135]. However, all these methods are facing and suffering from several problems, such as required a lot of external energy, high operating and maintenance costs, enzyme instability during their operations, long decomposition periods with low decolorization efficiency, and further sludge production which may needs further overcome.
It has been reported that MFCs has drawn an increasing interest in the simultaneous dye removal and electricity generation from various azo dye containing wastewater, such as congo red [133,136], methyl orange [91,137], methylene blue and methyl red [138], orange G [139], as well as methyl red [140]. Moreover, to increase performances, several practical applications were ensured for the operations of modified MFC configurations [141–143]. During the time of operations, MFC was exhibits better dye removal effect than conventional one [114,144]. Since the dye removal efficiency is highly depending up on anode nature which could be able to decompose a complex organic pollutant in to simpler one due to the formation of an active biofilm from bacterial enrichment. So, it has a significant factor to change the whole mechanism in MFCs during simultaneous decolorization and electricity generation [145]. So, the structural modification of conventional anode in the presence of EAM should be given the first priority to increase MFC performance [114]. Owing to this strategy, anode material with high electrical conductivity, best surface roughness, good stability, and low-cost have been met and provide promising criteria for dye removal in MFC [17]. As stated above, conventional carbon anode materials are the most widely used to date due to their low-cost, low content of heavy metal impurities, high quality, and environmentally friendly [21]. Their disposability is also the other criteria to use as a common electrode in MFC [23]. It was explained that conventional carbon anode have low efficiency towards bioelectricity and wastewater treatment [24]. If these factors are solved during operation, the anode material is then become a good functional material to carry a biofilm which act as a center for electron collection and a medium for electron transport. Therefore, the biofilm formation, resulting in increased catalytic activity followed by lowering in internal charge transfer resistance during substrate oxidation [45] is also one of the core concern in the bioelectrochemical system. Thus, surface modification of conventional carbon materials with urgent functional nanocomposite is one of the effective ways to achieve dye removal followed by current generation in MFC. Beside surface structure modification, the conductivity of the material could also reduce its ohmic resistance [25].
Combinations of redox polymers and inorganic NPs as a composite have attracted a considerable attention in many fields of applications [146] due to their facile synthesis, better electrical conductivity, environmental stability, fast electron transfer rate, excellent catalytic activity and reversible redox stability [45,46]. The effectiveness of nanocomposite conductivity leads to facilitate electron transfer rate and minimizes the electrochemical energy losses at the electrode/microorganism interface in the MFC device system [5,6,18]. So, incorporation of metal oxide nanostructures with conducting polymer matrix would promote an excellent catalytic activity towards various electrochemical reactions. So, in this review, such limitations were tried to discuss in brief and their possible modification techniques were reviewed. In general, scientific reports showed that, using an environmentally benign TMOs based nanocatalysts are a key and good indicator to remediate waste disposals beside their few drawbacks [147]. However, many factors play a major role in the anode performance of MFCs such as internal resistance, nature of catalyst, substrate concentration, and electrode spacing, electrode material properties, etc. [145].
Conclusion and future prospects
MFC is an electrochemical cell that enable to produce a renewable bioelectricity from renewable organic waste matters through the action of active exoelectrogenic bacteria and thus limit our dependence on hydrocarbon fuels. Such problem is a cause for cost ineffectiveness due to the slow electron transfer mechanism between electroactive microorganisms and anode surface. Creating an effective contact between active microbe and solid conductor through physical, chemical, electrochemical techniques play a vital role to enhance the performance of the cell device. To perform this, researchers have been primarily focused on low-cost conventional carbon electrode materials, but most of conventional carbon electrodes have poor chemical stability, small surface area, large pore size, poor mechanical stability, and low degree of biocompatibility. These electrodes show poor biocompatibility and limited electrocatalytic activity in the reactions at the biofilm/anode interface in MFC which limits both treatment efficiency and bioelectricity production. So, conventional carbon anodes need surface modification to reduce the ohmic resistance which is the dominant factor that affects MFC performances. Therefore, modifying these electrodes with functional hybrid composites derived from inorganic-organic integrations prepared by either biosynthesis (green) or chemical method. So, modification of conventional carbon-based anode with smart-functional nanomaterials composed from metal oxides and conjugated conducting polymers was effective to attain performances in MFC. Therefore, low-cost binary and ternary based composite modified anode will be expected for the future to commercialize MFC device improvement for bioremediation and its simultaneous power output.


